Given a barometric pressure of 762.4 mmHg, calculate the pressure of each gas sample as indicated by
Question:
Given a barometric pressure of 762.4 mmHg, calculate the pressure of each gas sample as indicated by the manometer.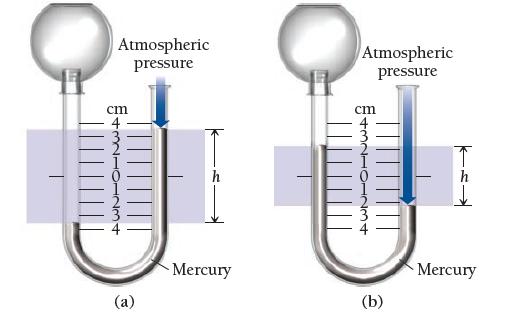
Transcribed Image Text:
Atmospheric pressure cm (a) Mercury Atmospheric pressure cm 11 (b) Mercury
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a 83...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Given a barometric pressure of 751.5 mmHg, calculate the pressure of each gas sample as indicated by the manometer. Atmospheric pressure cm (a) I Mercury Atmospheric pressure cm 4 MNOHY+ (b) Mercury
-
An 89.3 mL sample of wet O 2 (g) is collected over water at 21.3 C at a barometric pressure of 756 mmHg (vapor pressure of water at 21.3 C = 19 mmHg). (a) What is the partial pressure of O 2 (g) in...
-
(A) The reaction of aluminum with hydrochloric acid produces hydrogen gas. The balanced chemical equation for the reaction is given below. 2 Al(s) + 6 HCl(aq) 2 AlCl 3 (aq) + 3 H 2 (g) If 35.5 mL of...
-
How does the ERM system help with timely, effective, and democratic business decisions? A) Reporting to the presidents of each unit a faculty in a method that allows silo type decisions B) Board...
-
What is an enterprise system supposed to accomplish?
-
Alternative joint-cost-allocation methods, further-process decision. The Wood Spirits Company produces two productsturpentine and methanol (wood alcohol)by a joint process. Joint costs amount to...
-
Two conspirators, Timothy Carpenter and Timothy Sanders, were charged with aiding and abetting a robbery affecting interstate commerce and the use or carriage of a firearm. At trial, the FBI...
-
Best Trim, a manufacturer of lawn mowers, predicts that it will purchase 204,000 spark plugs next year. Best Trim estimates that 17,000 spark plugs will be required each month. A supplier quotes a...
-
DataBase managment design. Do the dowsings: 1. Run the sql commands written in ddl.sql file attached 2. Create_a dummy data. You can use "fake data generators" 3. Write followings - Write the query...
-
A sample of gas has an initial volume of 5.6 L at a pressure of 735 mmHg. If the volume of the gas is increased to 9.4 L, what is its pressure?
-
The North American record for highest recorded barometric pressure is 31.85 in Hg, set in 1989 in Northway, Alaska. Convert this pressure to each indicated unit. a. MmHg b. Atm c. Torr d. KPa...
-
Which standard deals with intangible non-current assets?
-
Why is the contractor normally required to submit a bid bond when making a proposal to an owner on a competitively bid contract?
-
Discuss what actions are necessary to make the integrated R&D network (or the transnational form) effective.
-
Highlight four conventional methods of market research, and define their strengths and weaknesses.
-
What is the role of TQM in innovation?
-
Why is the individual so important for an innovative organisation? What individual factors and considerations should be taken into account when managing an organisation for innovation?
-
Wallace Corporation summarizes the following information from its weekly payroll records during April. Prepare the two journal entries to record the payment of the payroll and the accrual of its...
-
The overall reaction and equilibrium constant value for a hydrogenoxygen fuel cell at 298 K is 2H 2 (g) + O 2 (g) 2H 2 O(l) K = 1.28 10 83 a. Calculate E cell and G 8 at 298 K for the fuel cell...
-
Determine the vertical displacement of joint D. Using Castiglianos theorem. AE is constant. Assume the members are pin connected at their ends. 000 4 m 4 m - 15 kN 20 kN 000
-
Determine the vertical displacement of joint D. Use the method of virtual work. AE is constant. Assume the members are pin connected at their ends. 000 4 m 4 m - 15 kN 20 kN 000
-
Determine the vertical displacement of joint E. For each member A = 400 mm 2 , E = 200 GPa. Using Castiglianos theorem. 1.5 m A JO 45 kN 2 m 2 m
-
You are the manager of a civil engineering pavement company that makes concrete for various highway projects in the State of Texas. Your company has various sites across the state to take sand and...
-
You are given the following information concerning Parrothead Enterprises: Debt: 9,400 6.6 percent coupon bonds outstanding, with 21 years to maturity and a quoted price of 105.00. These bonds have a...
-
A database developer normally uses several fact-finding techniques during a single database project. The five most commonly used techniques are examining documentation, interviewing, observing the...

Study smarter with the SolutionInn App


