Given that the H f of 1 M H 2 SO 3 is -633 kJ, use the
Question:
Given that the ΔH°f of 1 M H2SO3 is -633 kJ, use the data in Appendix II, Table B to calculate the ΔH° for the formation of a 1 M solution of SO2 in water from SO2(g).
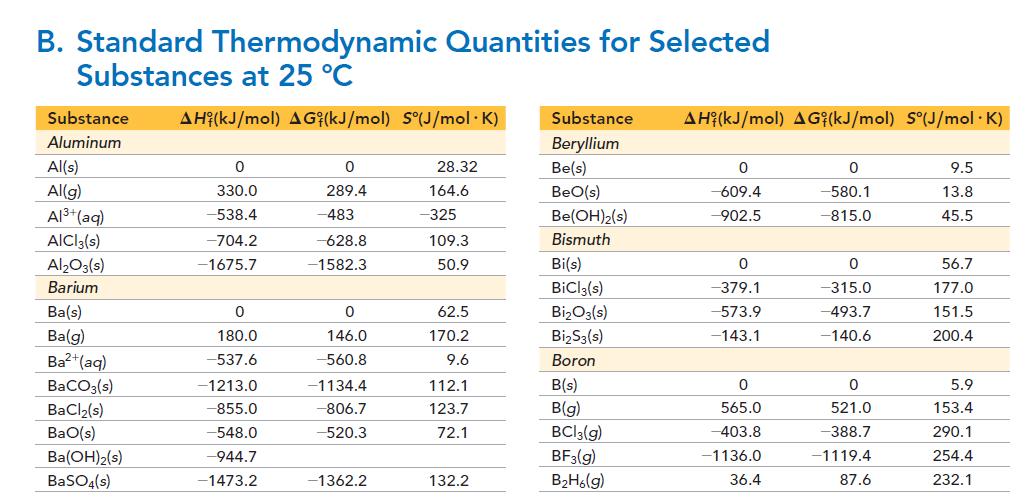
Transcribed Image Text:
B. Standard Thermodynamic Quantities for Selected Substances at 25 °C Substance AH (kJ/mol) AG (kJ/mol) S°(J/mol.K) Aluminum Al(s) Al(g) Al³+ (aq) AICI 3(s) Al₂O3(s) Barium Ba(s) Ba(g) Ba²+ (aq) BaCO3(s) BaCl₂(s) BaO(s) Ba(OH)2(s) BaSO4(s) 0 330.0 -538.4 -704.2 -1675.7 0 180.0 -537.6 -1213.0 -855.0 -548.0 -944.7 -1473.2 0 289.4 -483 -628.8 -1582.3 0 146.0 -560.8 -1134.4 -806.7 -520.3 -1362.2 28.32 164.6 -325 109.3 50.9 62.5 170.2 9.6 112.1 123.7 72.1 132.2 Substance AH (kJ/mol) AGi(kJ/mol) S°(J/mol .K) Beryllium Be(s) BeO(s) Be(OH)2(s) Bismuth Bi(s) BiCl3(s) Bi₂O3(s) Bi₂S3(s) Boron B(s) B(g) BC13(g) BF3(g) B₂H6(g) 0 -609.4 -902.5 0 -379.1 -573.9 -143.1 0 565.0 -403.8 -1136.0 36.4 0 -580.1 -815.0 0 -315.0 -493.7 -140.6 0 521.0 -388.7 -1119.4 87.6 9.5 13.8 45.5 56.7 177.0 151.5 200.4 5.9 153.4 290.1 254.4 232.1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Calculation of H rxn for Part I 0 1. Assume the heat capacity of the final solution is 4.184 J K-1 g-1. Using the final mass of the solution in the calorimeter, calculate q contents from equation...
-
Use the data in Appendix II, Table B to calculate H for the formation of a 1 M solution of H 2 SO 4 from SO 3 (g). B. Standard Thermodynamic Quantities for Selected Substances at 25 C Substance AH...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
In Exercises find the second derivative of the function. f(x) = 8 (x - 2)
-
Develop brief answers to each of the following questions: 1. In what way is selling an investment for a gain potentially a negative in evaluating quality of earnings? 2. Is it unethical for new...
-
Calculate the annual percentage rate of forgoing the cash discount under each of the following credit terms: a. 2/10, net 60 b. 2/10, net 30
-
Assume the same data as given in problem 9, except the company expects the following production: Case A: 300 bbl per month Case B: 500 bbl per month REQUIRED: a. Determine the number of months needed...
-
Marston Corporation manufactures disposable thermometers that are sold to hospitals through a network of independent sales agents located in the United States and Canada. These sales agents sell a...
-
2. Consider a consumer with CES utility u(x1, x2) = x + x2- (a) Does this consumer have preferences the "increasing difference prop- erty"? How about the strict increasing difference property? Make a...
-
Calculate the standard enthalpy of reaction for reducing the different forms of iron oxide to iron metal and CO 2 from the reaction of the oxide with CO. Identify which reaction is the most...
-
Breathing air that contains 0.13% CO by volume for 30 minutes will cause death. CO can form by incomplete combustion of carbon-containing compounds. Calculate the minimum volume of octane (C 8 H 18 ,...
-
Determine whether recording each of the following adjustments will increase (I), decrease (D), or have no effect (NE) on each of the three elements of the accounting equation....
-
Problem 3. Consider the following context-free grammar: G -> GB G - GN G-> A B-> (E) EE (E) E-> A N (L] LLE L-> L ( L -> A a. Express the language of strings generated by the above grammar.
-
Moore Electric has sales of $3,800,000. Variable costs are $2,130,000 and controllable fixed costs are $782,000. The company's average operating assets are $5,000,000. If the company decreases their...
-
A new project has an initial cost of $148,000. The equipment will be depreciated on a straight-line basis to a book value of $49,000 at the end of the four-year life of the project. The projected net...
-
Your company is about to acquire a small firm whose growth has been about 35% per year. Quite a nice catch. The acquired firm has been strong in its particular market. Their financial statements have...
-
What does the abbreviation CPC stand for? Explain.
-
How does global human resource management differ from domestic human resource management?
-
Synthesize the products by drawing out reagents and intermediates along the way. `N H. OH HO HO
-
A long, straight wire carries current I = 100 A in a region where the magnetic field has a magnitude B = 10 T, but it is found that the force on the wire is zero. Explain how that can be.
-
A long, straight wire of length 0.75 m carries current I = 1.5 A in a region where B = 2.3 T. If the force on the wire is 1.4 N, what is the angle between the field and the wire?
-
Repeat Problem 47, but assume the force is 5.6 N. Is this possible? Data from Problem 47 A long, straight wire of length 0.75 m carries current I = 1.5 A in a region where B = 2.3 T. If the force on...
-
1. To measure the understanding level on knowledge about ethical framework of business environment a. Steps to minimize Workplace Bullying b. Measures to avoid Sexual Misconduct or Harassment c....
-
We represent a client who is a combat veteran. He is a patient of Jane Jones, a psychiatrist. He sent a medical record request to Dr. Jones, and she gave him records that were clearly redacted (with...
-
Respond to the assignment topic completely.Cite the particular MD Attorneys' Rules of Professional Conductfor authority to your response. ABA Model Rules and paralegal association ethics rules are...

Study smarter with the SolutionInn App


