In each reaction, identify the BrnstedLowry acid, the Brnsted Lowry base, the conjugate acid, and the conjugate
Question:
In each reaction, identify the Brønsted–Lowry acid, the Brønsted– Lowry base, the conjugate acid, and the conjugate base.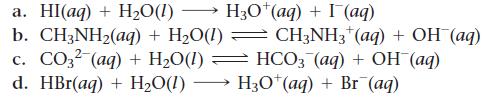
Transcribed Image Text:
H30¹(aq) + (aq) a. HI(aq) + H₂O(1)→ b. CH3NH₂(aq) + H₂O(1) CH3NH3 (aq) + OH (aq) c. CO32 (aq) + H₂O(1) — HCO3 (aq) + OH (aq) d. HBr(aq) + H₂O(1) → H3O+ (aq) + Br (aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 25% (4 reviews)
In each reaction in the image you sentthe BrnstedLowry acid is the speci...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
An investor wishes to analyse the effects of different compounding frequencies Suppose 1000 is invested for 1 year at an interest rate of 5 per annum compounded Assume there are 365 days in 1 year
-
If 100 mol methanol and 100 mol acetic acid are fed to a batch reactor and allowed to come to equilibrium. How many moles of each component will be present in the product stream?
-
The conjugate base of diethyl malonate can serve as a nucleophile to attack a wide range of electrophiles. Identify the product that is formed when the conjugate base of diethyl malonate reacts with...
-
During 2018, Susan incurred and paid the following expenses for Beth (her daughter), Ed (her father), and herself: Surgery for...
-
Serene Assurance Company provides both automobile and life insurance. The projected income statements for the two products are as follows: The president of the company is considering dropping the...
-
The highlow method of analyzing mixed costs uses only two observation points: the high and the low points of activity. Are these always the best points for prediction purposes? Why or why not?
-
What needs to be contained in the caption of a complaint?
-
Before maturity, Foster Incorporated sold $500,000 of 12% bonds on January 1, 2013, for $470,143.47, a price that yields a 14% interest rate. The bonds pay interest semiannually on June 30 and...
-
Azhan started his business in 2013 as a sole trader, registered under the name of Nahza Trading (accounting year end 31 December). In 2016 he got married, and in 2017 Azhan set up a company as an...
-
Write the formula for the conjugate base of each acid. a. HCI b. HSO3 c. HCHO, d. HF
-
In each reaction, identify the BrnstedLowry acid, the Brnsted Lowry base, the conjugate acid, and the conjugate base. a. HCO3(aq) + HO(1) b. NH3(aq) + HO(1) c. HNO3(aq) + HO(1) H3O+ (aq) + NO3 (aq)...
-
Sketch the p.d.f. of the 2 distribution with m degrees of freedom for each of the following values of m. Locate the mean, the median, and the mode on each sketch. (a) m = 1. (b) m = 2. (c) m = 3. (d)...
-
3. Provide examples of how the current and future banking regulations will impact the industry both positively and negatively.
-
How do taxonomists address the taxonomic inflation problem, particularly concerning the proliferation of species concepts and the impact on biodiversity conservation efforts ? Explain
-
What is the "Fischer equation" and what use would this equation have for capital budgeting? Typically, annual depreciation allowances are not indexed to inflation. What is the impact of inflation on...
-
When considering the growth differential between GM and Tesla, which model for valuation might be more prone to error when trying to value Tesla (Free Cashflow vs Earnings)? Please explain.
-
Maxim paid $5,162.94 for a $5,000 face value bond bearing interest at 6.2% payable semiannually. The bond has 8 years remaining to maturity. What is the bond's yield to maturity? (Round your final...
-
Following are the balance sheet accounts of a small business: Cash..................................................... $ 5,000 Accounts Payable................................ ?...
-
Solve each equation or inequality. |6x8-4 = 0
-
A 3/4-in plastic swing check valve carries 18 gal/min of seawater at 77F. Compute the expected pressure drop across the valve.
-
A 3-in plastic swing check valve carries 300 gal/min of kerosene at 77F. Compute the expected pressure drop across the valve.
-
An 8-in plastic swing check valve carries 3500 gal/min of glycerin at 77F. Compute the expected pressure drop across the valve.
-
my h 112 m, = 20 kg m = 30 kg h=2.5 m r = 1.50 m Cart 1 collides with cart 2. Cart 1 stops and cart 2 continues through the loop (they do NOT stick) a. What is the speed of cart 2 after the...
-
Carla Vista Company has the following year-end account balances on November 30, 2024: Service Revenue $39,000; Insurance Expense $2,500; Rent Expense $7,200; Supplies Expense $1,750; L. Lee, Capital...
-
Roger Ltd. commences operations on 1 July 2018. On the same date, the company purchases a machine for $900 000. The expected useful life of the machinery is four years. The machinery has no residual...

Study smarter with the SolutionInn App


