Use formal charge to identify the better Lewis structure. H H-C= H H=C
Question:
Use formal charge to identify the better Lewis structure.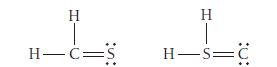
Transcribed Image Text:
H H-C= H H=C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
H HCS 00 ...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A long conducting wire made of anti-matter carries a uniform current per unit area. The anti-electrons (each carries charge +1.602 x 10-19 C ) with speed 4.862 m/s (the speed and magnitude of the...
-
Use formal charges to identify the better Lewis structure. H H-S-C-H 1 H H T H-C-S-H I H
-
LMO Company incurred expenditures for leasehold improvements during the year. The relevant lease is on an office building that the Company rented at the beginning of 2021. The improvements cost...
-
Consider the many moments of joy in the movie. Why include them? What risky acts of courage do Katherine, Mary and Dorothy take? How are they rewarded? The film shows the strong relationships that...
-
What is online analytical processing? How is OLAP related to databases? What is a pivot table, and how are pivot tables and OLAP related?
-
Refer to the financial statements for Rusco Products in Problem 1511. Because the Cash account decreased so dramatically during 2009, the companys executive committee is anxious to see how the income...
-
In 2001, the City of New York and the Dormitory Authority of the State of New York (DASNY) entered into an agreement to build a forensic biology laboratory in Manhattan. Per the agreement, DASNY...
-
a. Create a network flow model for this problem. (Consider inserting intermediate nodes in your network to assist in meeting the minimum monthly purchase restrictions for each supplier and the...
-
The demand curve for cookies is a rightward curve and the quantity demanded is 100 when the price of cookies is $2.00. What happens to consumer surplus when the price is $3.00? What happens to...
-
In N 2 O, nitrogen is the central atom, and the oxygen atom is terminal. In OF 2 , however, oxygen is the central atom. Use formal charges to explain why. Formal charge = number of valence electrons...
-
Write a Lewis structure that obeys the octet rule for each ion. Include resonance structures if necessary and assign formal charges to each atom. a. CIO3 b. ClO4 c. NO3 + d. NH4
-
Penny Davis runs the Shear Beauty Salon near a college campus. Several months ago, Penny used some unused space at the back of the salon and bought two used tanning beds. She hired a receptionist and...
-
Suppose your model predicts that overweight children have an 80% higher risk of suffering from diabetes in their adult life. If data show that overweight children do not suffer from diabetes as...
-
Suppose that in a given economy all goods and services produced are sold in perfectly competitive markets. Would you represent this economy using the classical or Keynesian approach? Explain why.
-
Suppose Japan has a GDP of $5 trillion, and that its national savings rate is 25%. Assuming Japan is an open economy, a) calculate Japans investment if net exports are 1% of GDP. b) calculate Japans...
-
Social media firms strategy has three pillars: (1) keeping user addicted to the content, (2) collecting data about user behavior, (3) selling advertisements targeting specific users. Do you really...
-
Comment on the effect of a decrease in autonomous investment on wealth when the economy can be considered a a) closed economy. b) small open economy.
-
Go through each of the arguments for restricting trade (both those of general validity and those having some validity for specific countries) and provide a counterargument for not restricting trade.
-
Suppose you need to answer any four of seven essay questions on a history test and you can answer them in any order. a. How many different question combinations are possible? b. What is the...
-
What is the barometric pressure reading in millimeters of mercury corresponding to 101.325 kPa(abs)?
-
Why must a barometric pressure reading be corrected for temperature?
-
By how much would the barometric pressure reading decrease from its sea-level value at an elevation of 1250 ft?
-
Relevance has always been the challenge of the advertising industry. Summarize Kristi Roger's assessment of the advertising industry in her TED Talk. Considering her hypothesis and results, what are...
-
What are some examples of things you can't do on company computers and why?
-
What is a good example of Strategy Implementation - including timeline for a psychiatric hospital that is planning to add an additional service?

Study smarter with the SolutionInn App


