Name each alkene. a. CH3 CH -CH=CH-CH - CH3 b. CH3-CH-CH=CH-CH3 I CH3 CH3 T CH T
Question:
Name each alkene.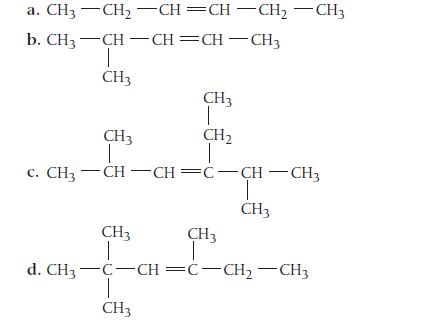
Transcribed Image Text:
a. CH3 CH₂ -CH=CH-CH₂ - CH3 b. CH3-CH-CH=CH-CH3 I CH3 CH3 T CH₂ T C. CH3—CH…CH=C-CHCH3 CH3 I CH3 CH3 d. CH3-C-CH=C-CH₂CH3 T CH3 CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
CH3CH2CHCH CH CH3 a b CH3 I CH3 CHCH...View the full answer

Answered By

Pranav Makode
I am a bachelor students studying at professor ram meghe institute of technology and research. I have a great experience of being an expert. I have worked as an expert at helloexperts and solvelancer as a part time job. I have also worked as a doubt solver at ICAD SCHOOL OF LEARNING, which is in Amravati city. I have also worked as an Freelancer.
I have great experience of helping students, as described above. I can help any students in a most simple and understandable way. I will not give you have any chance for complaint. You will be greatfull to accept me as an expert.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Write a structural formula or build a molecular model and give a correct IUPAC name for each alkene of molecular formula C7H14 that has a tetrasubstituted double bond.
-
Give an example reaction that would yield the following products. Name the organic reactant and product in each reaction. a. Alkane b. Monohalogenated alkane c. Dihalogenated alkane d. Tetra ha...
-
Write a structural formula or build a molecular model and give a correct IUPAC name for each alkene of molecular formula C7H14 that has a tetra substituted double bond.
-
Paula makes the following acquisitions of ordinary shares in Indigent plc: She sells 500 shares on 28 March 2021. No shares are acquired within the next 30 days. (a) Show the s104 holding on 28 March...
-
Marie Companys balance sheet shows total liabilities of $678,000, total equity of $226,000, and total assets of $904,000. Required: Round answers to two decimal places. 1. Calculate the debt ratio...
-
In this chapter a decision tree was developed for John Thompson (Figure for the complete decision tree analysis). After completing the analysis, John was not completely sure that he is indifferent to...
-
If the IRR of Alternative \(A\) is 18 percent, the IRR of Alternative B is 16 percent, and MARR is 12 percent, which of the following is correct? a. Alternative B is preferred over Alternative A. b....
-
Doughty Sawmill, Inc., (DSI) purchases logs from independent timber contractors and processes the logs into three types of lumber products: Studs for residential buildings (walls, ceilings)...
-
Problem 4. Consider the following imperfect information game. Notice that both play- ers have two information sets. 5,2 1 a b 2 t 1 d d 4,3 6.0 4,6 0,4 6,0 (a) Find all subgame perfect Nash...
-
Write structural formulas for each of the possible isomers of n-hexene that are formed by moving the position of the double bond.
-
Name each alkene. a. CH,=CHCH,CH, CH3 CH3 T T CH3-CH-C=CH-CH3 CH,=HCCHCH,CH, CH3 b. c. CH3 CH I CH3 CH3 I d. CH3-CH-CH=C-CH3 CH - CH3
-
List and describe the three major categories of business reports.
-
A company has made following estimates if the CFAT of the proposed project. The company uses decision tree analysis to get clear picture of project's cash inflow. The project cost is 1,50,000 and the...
-
Your company plans to produce a new product, a wireless computer mouse. Two machines can be used to make the mouse, Machines A and B. The price per mouse will be $23.00 regardless of which machine is...
-
Over each of the next 4 years, McDougall Corp. expects to sell 56,400 units of its product at a price of $6.00 per unit. Annual fixed costs are expected to be $18,100 and variable costs are estimated...
-
A corporation is 100% owned by an individual. In 2023, it reports business gross receipts of 100,000, interest income of 200,000, and capital gain of 300,000. Assume it has no expenses. WHAT IS THE...
-
The approval authority for O&M construction projects in excess of $750,000, but less than $2,000,000: a. Under Secretary of Defense or designee b. Assistant Secretary of the Army for Acquisition,...
-
Construct a cause-and-effect diagram to reflect student dissatisfied with university registration process. Use the four Ms or create your own organizing scheme. Include at least 12 causes.
-
Synthesize the products by drawing out reagents and intermediates along the way. `N H. OH HO HO
-
Vertically polarized light passes through a polarizer with its axis at 70 to the vertical. What fraction of the incident intensity emerges from the polarizer?
-
A typical laboratory electric field is 1500 V/m. Find the average intensity of an electromagnetic wave with this value for its peak field.
-
What would be the average intensity of a laser beam so strong that its electric field produced dielectric breakdown of air (which requires Ep = 3 MV/m)?
-
Trevor, a single taxpayer, received $170,000 in wages from his employer through November 30, 2023. On December 1, 2023, the employer pays Trevor a $50,000 bonus. Prior to December 1, the employer was...
-
15. Eloise plans to accumulate 50,000 at the end of 40 years. She makes the following deposits: (i) X at the beginning of years 1 - 15; (ii) No deposits at the beginning of years 16 - 25; (iii) Y at...
-
Peer group analysis is a method used to evaluate a company's performance by comparing it to a group of similar businesses. Imagine it as an "apples-to-apples" comparison, where you're looking at...

Study smarter with the SolutionInn App


