Name each alkene. a. CH,=CHCH,CH, CH3 CH3 T T CH3-CH-C=CH-CH3 CH,=HCCHCH,CH, CH3 b. c. CH3 CH I
Question:
Name each alkene.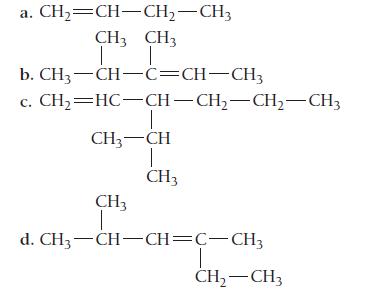
Transcribed Image Text:
a. CH,=CH–CH,—CH, CH3 CH3 T T CH3-CH-C=CH-CH3 CH,=HC—CH–CH,—CH, CH3 b. c. CH3 CH I CH3 CH3 I d. CH3-CH-CH=C-CH3 CH₂ - CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
a 1butene b 34...View the full answer

Answered By

Lokesh Singh
I'm an IT professional with expertise in Cybersecurity, Sysadmin, MS Windows, Linux, and DevOps MS Office and Network Administration. With over 3 years of experience in the IT industry, I am highly knowledgeable in the latest technologies and trends.
I am an expert in developing and managing innovative solutions to complex problems and have a proven track record of success. I am also an effective communicator and have excellent interpersonal and organizational skills. I take great pride in my work and strive to provide the best results for every project. I'm always looking for new opportunities to further my knowledge in the technology field and I'm excited to see what the future holds.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Write a structural formula or build a molecular model and give a correct IUPAC name for each alkene of molecular formula C7H14 that has a tetrasubstituted double bond.
-
Give an example reaction that would yield the following products. Name the organic reactant and product in each reaction. a. Alkane b. Monohalogenated alkane c. Dihalogenated alkane d. Tetra ha...
-
Write a structural formula or build a molecular model and give a correct IUPAC name for each alkene of molecular formula C7H14 that has a tetra substituted double bond.
-
Paul made the following acquisitions and disposals of ordinary shares in Crimmon plc: No further shares were acquired during 2020. Against which acquisitions will each of the above disposals be...
-
Groomer Company provided the following income statement for last year: Sales ...................$1,400,000 Cost of goods sold .................550,000 Gross margin ................$ 850,000 Operating...
-
Coren Chemical, Inc., develops industrial chemicals that are used by other manufacturers to produce photographic chemicals, preservatives, and lubricants. One of their products, K-1000, is used by...
-
What do you know about the mathematical value of a project's internal rate of return under each of the following conditions? a. The present worth of the project is greater than 0 . b. The present...
-
Santana, Inc. reports the following liabilities (in thousands) on its January 31, 2014, balance sheet and notes to the financial statements. Instructions (a) Identify which of the above liabilities...
-
On a private flight with capacity for 150 people, $800,000 per person is charged, plus $25,000 for each unsold seat on the plane. How many people must travel on the plane and what will be the price...
-
Name each alkene. a. CH3 CH -CH=CH-CH - CH3 b. CH3-CH-CH=CH-CH3 I CH3 CH3 T CH T C. CH3CHCH=C-CHCH3 CH3 I CH3 CH3 d. CH3-C-CH=C-CHCH3 T CH3 CH3
-
List all the possible products for each alkane substitution reaction. a. CH4 + Cl b. CH3CHBr + Br C. CH3CHCHCH3 + Cl d. CH3CHBr2 + Br
-
Danielle Anderson, your client and a cash method taxpayer, works full-time at a music store located in a mall. She assists the manager in buying decisions, serves customers on the sales floor, and...
-
2. How does the ITAA define 'trading stock'? Give examples of assets that could constitute 'trading stock' of a business. 2.1Under Division 70 ITAA 1997, concisely outline how a taxpayer must go...
-
Analyze the quarterly earnings per share (EPS) announcement on 3 May 2018 for ESS-US (FactSet identifier). Was this a positive, neutral, or negative earnings surprise? Positive Neutral Negative Mark...
-
Analyze how critical infrastructure became a component of the national preparedness system and mission planning frameworks.
-
AC/DC Corporation (*the Company" currently manufactures 72,000 optical switches that it uses in several of its hnal products. Management is considering whether to continue manufacturing the optical...
-
You are an analyst for XYZ Real Estate Inc. The vice- president has asked you to perform a basic analysis on an industrial property that the company is considering acquiring. The vice-president is...
-
Consider the everyday task of getting to work on time or arriving at your first class on time in the morning. Draw a fish-bone chart showing reasons why you might arrive late in the morning.
-
You have just begun your summer internship at Omni Instruments. The company supplies sterilized surgical instruments for physicians. To expand sales, Omni is considering paying a commission to its...
-
Estimate the peak electric field inside a 1.1-kW microwave oven under the simplifying approximation that the microwaves propagate as a plane wave through the ovens 750cm 2 cross-sectional area.
-
Your new radio says it can pick up signals with peak electric fields as weak as 450V/m. Will it work if you take it to your remote cabin, where the intensity of your favorite radio station is 0.35...
-
A laser pointer delivers 0.10-mW average power in a beam 0.90mm in diameter. Find (a) the average intensity, (b) the peak electric field, (c) the peak magnetic field.
-
At the present time, Western Gas & Electric Company (WGC) has 10-year noncallable bonds with a face value of $1,000 that are outstanding. These bonds have a current market price of $1,092.79 per...
-
A global corporation that fabricated financial records to increase sales and mislead investors was implicated in a recent accounting controversy. This unlawful behavior was revealed by a...
-
To get things started, or pointed in the right direction Slide 4: Recommend a budget type appropriate for the scenario and describe the benefit that will result from this budget type. The Vegan...

Study smarter with the SolutionInn App


