Name each compound in which the benzene ring is best treated as a substituent. CH3 | a.
Question:
Name each compound in which the benzene ring is best treated as a substituent.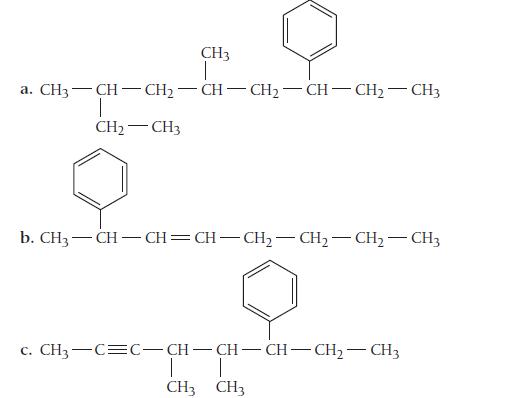
Transcribed Image Text:
CH3 | a. CH3—CH=CH-CH-CH=CH-CH2–CH3 T CH₂CH3 b. CH,—CH—CH=CH–CH,—CH,—CH, CH3 c. CH,—C=C–CH-CH-CH–CH,—CH3 T CH3 CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
a 35dimethyl7...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Name each compound in which the benzene ring is best treated as a substituent. a. H3C-CH-CH-CH-CH-CH3 H3C b. CH,CH,CH,CH,C=CCH3 CH3 | c. CH3CH=CH-C=CHCHCH,CH3 T CH3 CH3
-
Give the product of the following reaction: If the terminal sp2 carbon of the substituent bonded to the benzene ring is labeled with 14C, where will the label be in the product? A
-
Arene C has the composition 90.6% carbon and 9.4% hydrogen. Its mass and 1H NMR spectra are shown below. a. b. a. Calculate the empirical formula of C. b. From the mass spectrum, find the molecular...
-
In the year to 5 April 2021, Thomas More made the following disposals: (i) A flat in a house that he had purchased on 1 December 2010 for 80,000. It had never been occupied as the main residence and...
-
Refer to the Kepler Company information as shown in Problem 16-45. Required: 1. Compute the following for each year: a. The times-interest-earned ratio b. The debt ratio 2. Does Kepler have too much...
-
Allen Young has always been proud of his personal investment strategies and has done very well over the past several years. He invests primarily in the stock market. Over the past several months,...
-
Consider the following cash flow profile and assume MARR is 10 percent/year. a. What does Descartes' rule of signs tell us about the IRR(s) of this project? b. What does Norstrom's criterion tell us...
-
Flexible budget, working backward. The Clarkson Company produces engine parts for car manufacturers. A new accountant intern at Clarkson has accidentally deleted the calculations on the companys...
-
= Suppose f(z) is analytic for |z| < 3. If |(z)| 1, and (i) (1) = 0, what is the maximum value of |f(0)|? For which func- tions is the maximum attained?
-
Name each disubstituted benzene. a. Br Br b. CH - CH3 CH - CH3 C. F
-
Name each monosubstituted benzene. a. HC-CH3 b. F C. CH3 H3C-C-CH3
-
What is the key financial innovation that has greatly facilitated the growth of Islamic-style asset management?
-
A farmer has a rectangular field with dimensions 120 meters by 80 meters. He wants to build a fence around the field to divide it into two equal parts. What is the minimum length of the fence he...
-
The unadjusted trial balance of Farmer Investment Advisers at December 3 1 , 2 0 1 6 , follows: ( Click the icon to view the unadjusted trial balance. ) Adjustment data at December 3 1 , 2 0 1 6 :...
-
Analyzing a Writing Scenario: Improving a Poorly Crafted Memo The following memo is poorly designed and formatted. In addition, it contains awkward wording and typographical errors, and it does not...
-
A car rental company charges a base fee of $50 plus $0.25 per mile driven. If John rented a car and paid $125 in total, how many miles did he drive?
-
Remember, a bonds coupon rate partially determines the interest-based return that a bond (will / might) pay, and a bondholders required return reflects the return that a bondholder (is obligated /...
-
What effect does insurance have on risk?
-
From the choice of simple moving average, exponential smoothing, and linear regression analysis, which forecasting technique would you consider the most accurate? Why? please write it in word...
-
A damped LC circuit consists of a 0.15F capacitor and a 20-mH inductor with resistance 1.6 . How many oscillation cycles will occur before the peak capacitor voltage drops to half its initial value?
-
A damped RLC circuit includes a 5.0 resistor and a 100-mH inductor. If half the initial energy is lost after 15 cycles, whats the capacitance?
-
An RLC circuit includes a 1.5-H inductor and a 250F capacitor rated at 400 V. The circuit is connected across a sine-wave generator with V p = 32 V. What minimum resistance will ensure that the...
-
9. At time t = 0, Paul deposits P into a fund crediting interest at an effective annual interest rate of 6.9%. At the end of each year in years 12 through 31, Paul withdraws an amount sufficient to...
-
How can this phenomenon be explained using the Heckscher-Ohlin model? "Singapore in 1965 was a low-skill labour abundant country compared to the rest of the world and exported low skill labour...
-
Writing an informal Business Report The Scenario The medium size tech company that employs you provides advisory and professional services. In consideration of the trend toward remote work they...

Study smarter with the SolutionInn App


