Nomex, a condensation copolymer used by firefighters because of its flame-resistant properties, forms from isophthalic acid and
Question:
Nomex, a condensation copolymer used by firefighters because of its flame-resistant properties, forms from isophthalic acid and m-aminoaniline. Draw the structure of the dimer.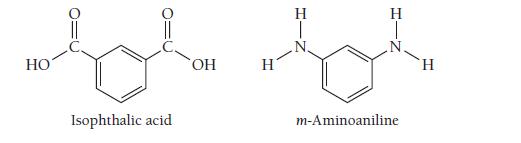
Transcribed Image Text:
HO Isophthalic acid OH H H T H m-Aminoaniline H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
Chemical reaction between isophthal...View the full answer

Answered By

Vikash singh
i would like to solve the problems starting from basic to higher level.
Everything question having logic i would like to explore that logic and solve the question in short way in short time.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The polyester named Lactomer® is an alternating copolymer of lactic acid and glycolic acid. Lactomer is used for absorbable suture material because stitches of Lactomer hydrolyze slowly over a...
-
Draw the condensed structure of the compounds formed by condensation reactions between (a) benzoic acid and ethanol, (b) ethanoic acid and methylamine, (c) acetic acid and phenol. Name the compound...
-
The total pressure of vapor over liquid acetic acid at 71.3°C is 146 mmHg. If the density of the vapor is 0.702 g/L, what is the mole fraction of dimer in the vapor? See Problem 11.149. Problem...
-
Jansen, Inc., is a defense contractor that uses job costing. Because the firm uses a perpetual inventory system, the three supporting schedules to the income statement (the schedule of raw materials...
-
First Development Corporation of Kentucky (FDCK) sought to purchase a fifteen-acre parcel of riverfront property owned by Martin Marietta. On May 9, FDCK made an offer to purchase the property for...
-
To test whether sales have been recorded, a sample should be drawn from a file of; a. Purchase orders. b. Sales orders. c. Sales invoices. d. Bills of lading. Choose the correct answer.
-
The pipe lies in the horizontal plane. If it is subjected to a vertical force \(\mathbf{P}\) at its end, determine the strain energy due to bending and torsion. Express the results in terms of the...
-
On August 1, 2016, the accountant for Western Imports downloaded the company's July 31, 2016, bank statement from the bank's website. The balance shown on the bank statement was $28,760. The July 31,...
-
How does the psychologic and economic impact differ from other forms of terrorism such as cyber terrorism? Also, how does a client's prejudices and attitudes can pose a danger to the personal...
-
Polyacetylene is an addition polymer with the structure shown here. Draw the structure of the monomer. H [] C. C H
-
One kind of polyester is a condensation copolymer formed from terephthalic acid and ethylene glycol. Draw the structure of the dimer. HO Terephthalic acid OH HD-CH-CH-OH Ethylene glycol
-
During the year, Xenoc produces 1,200 pairs of speakers and sells 1,000 pairs. Required What is net income using full costing?
-
Astro Company sold 20,000 units of its only product and reported income of $25,000 for the current year. During a planning session for next year's activities, the production manager notes that...
-
How do genomic imprinting and parent-of-origin effects shape gene expression patterns, and what are the implications for diseases associated with disruptions in imprinting, such as Prader-Willi and...
-
Ronald Powers, a well-known local entrepreneur, owned several businesses that filed for bankruptcy from 2010 to 2015. Last year, Powers established R. P. Properties, Inc. Powers is the sole...
-
Please view the video and think through what these supply chains may look like if these technologies or practices become ubiquitous. Let me know your thoughts within 500 word limit in the context of...
-
Explain the concept of gene-environment interactions, and how these interactions influence the expression of complex traits and the risk of developing multifactorial diseases ?
-
Why are direct materials price variances usually recorded at the time of purchase?
-
Draw the major product for each of the following reactions: (a) (b) (c) 1) 9-BBN 2) H2O2, NaOH 1) Disiamylborane 2) H20, NaOH
-
The three lowest energy levels for atomic carbon (C) have the following energies and degeneracies: What is the contribution to the average molar energy from the electronic degrees of freedom for C...
-
The lowest four energy levels for atomic vanadium (V) have the following energies and degeneracies: What is the contribution to the average energy from electronic degrees of freedom for V when T =...
-
Consider the following table of diatomic molecules and associated rotational constants: a. Calculate the rotational temperature for each molecule. b. Assuming that these species remain gaseous at 100...
-
(5) Evaluate the following integrals (a)]. 23+2x -da x+4x2+7" (1+3lnx) dr (b)/(1+3 T (c)/dr +5
-
1. Below is selected information from the yearly financial statements of Dias Corporation: 2022 2021 Total assets $500,000 $507,500 Total liabilities 408,850 395,500 Interest expense 30,000 20,250...
-
Factor. v + 10v+25

Study smarter with the SolutionInn App


