Palladium forms three different compounds with sulfur. The mass of sulfur per gram of palladium in each
Question:
Palladium forms three different compounds with sulfur. The mass of sulfur per gram of palladium in each compound is listed here. Show that these masses are consistent with the law of multiple proportions.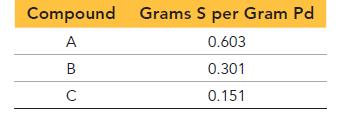
Transcribed Image Text:
Compound Grams S per Gram Pd A 0.603 B 0.301 C 0.151
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
The law of multiple proportions states that when two elements form a series of compounds the differe...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Sulfur and fluorine form several different compounds including sulfur hexafluoride and sulfur tetrafluoride. Decomposition of a sample of sulfur hexafluoride produces 4.45 g of fluorine and 1.25 g of...
-
In a series of experiments, a chemist prepared three different compounds that contain only iodine and fluorine and determined the mass of each element in each compound: (a) Calculate the mass of...
-
Sulfur and oxygen form both sulfur dioxide and sulfur trioxide. When samples of these are decomposed, the sulfur dioxide produces 3.49 g oxygen and 3.50 g sulfur, while the sulfur trioxide produces...
-
Comprehensive variance analysis review. Sonnet Inc. has the following budgeted standards for the month of March 2010: Sales of 2,000,000 units are budgeted for March. Actual March results are: Unit...
-
Austin Enterprises makes and sells three types of dress shirts. Management is trying to determine the most profitable mix. Sales prices, demand, and use of manufacturing inputs follow: The company...
-
The airline industry was hit particularly hard after the 9/11 attacks on the World Trade Center in 2001. In 2002, Southwest Airlines, one of the healthier airline companies, made a decision to...
-
The December 1, bank statement of Billys Hamburger just arrived from Safety Bank. To prepare the bank reconciliation, you gather the following additional data: a. The following checks are outstanding...
-
Lytton Ltd. uses a normal job-costing system with two direct cost categories (direct materials and direct labour) and one indirect cost pool. It allocates manufacturing overhead to jobs using a...
-
As part of a quality-assurance program being initiated by Community Hospital, each vice president personally interviewed every staff member under his or her jurisdiction to determine what actions...
-
In a process development study on yield, four factors were studied, each at two levels: time (A), concentration (B), pressure (C), and temperature (D). A single replicate of a 2 4 design was run, and...
-
Upon decomposition, one sample of magnesium fluoride produces 1.65 kg of magnesium and 2.57 kg of fluorine. A second sample produces 1.32 kg of magnesium. How much fluorine (in grams) does the second...
-
The mass ratio of sodium to fluorine in sodium fluoride is 1.21:1. A sample of sodium fluoride produces 28.8 g of sodium upon decomposition. How much fluorine (in grams) forms?
-
The demand for airline travel is quite sensitive to price. Typically, there is an inverse relationship between demand and price; when price decreases, demand increases, and vice versa. One major...
-
Describe the similarities and differences between periodic and perpetual inventory accounting information systems. Include in your discussion the impacts on the types of tests performed and audit...
-
The CEO of Textrala Petroleum, a crude oil wholesaler, is on an incentive compensation plan that weighs accounting net income heavily when awarding compensation. For 2003, Textrala has done so well...
-
Describe why using confirmations for testing completeness of accounts payable is more challenging than using confirmations for testing existence of accounts receivable? How can auditors address this...
-
Examine the following questions from an internal control questionnaire for the assembly process of a yacht manufacturer. Assume that the answer to each of these is "no" (which means that the control...
-
Describe the inventory price test, explicitly separating how the auditor collects evidence to support that the appropriate cost of inventory and to support the appropriate market value of inventory....
-
Medical Distributors, Inc. is a U.S. company that buys and sells used medical equipment throughout the United States and Canada. During the month of June, the company had the following transactions...
-
The following T-accounts show postings of selected transactions. Indicate the journal used in recording each of these postings a through e. Cash Accounts Receivable Inventory (d) 500 (e) 300 (b)...
-
Fill in the missing products below. excess HI Heat 1) Hg(OAc), Et 2) NABH, -: Na MCPBA 1) NaSH 2) H20 HBr
-
Propose a plausible synthesis for each transformation. a. b. c. d. e. f. g. h. i. j. k. l. m. n. o. p. q. r. s. t. u.
-
Propose a structure for ether with molecular formula C 7 H 8 O that exhibits the following 13 C NMR spectrum. Carbon NMR 114.0 129.5, 120,714.0 55.1- 159.71 160 120 140 100 100 Chemical shift (ppm)...
-
Risk levels are incorporated into potential investments by using discount rates for the level of risk associated with the specific investment ( Smart & Zutter, 2 0 2 2 ) . The riskier the investment,...
-
This case examines the October 2 0 1 5 initial public offering pricing decision for legendary Italian sports car company Ferrari by Fiat Chrysler management. We will evaluate Ferrari in light of...
-
You are thinking about buying BNS (Bank of Nova Scotia) stock. Their stock is currently trading for $81/share and pays and annual dividend of $4.12. You want to earn at least 8% on this investment in...

Study smarter with the SolutionInn App


