Rank each set of substances in order of increasing standard molar entropy (S). Explain your reasoning. a.
Question:
Rank each set of substances in order of increasing standard molar entropy (S°). Explain your reasoning.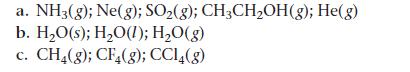
Transcribed Image Text:
a. NH3(g); Ne(g); SO₂(g); CH3CH₂OH(g); He(g) b. H₂O(s); H₂O(1); H₂O(g) C. CH4(g); CF4(g); CC14(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
a He Ne SO 2 NH 3 CH 3 CH 2 OH From He to Ne there is an ...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Rank each set of substances in order of increasing standard molar entropy (S). Explain your reasoning. a. 1(g); F(g); Br2(g); Cl(g) b. HO(g); HO(g); HS(g) c. C(s, graphite); C(s, diamond); C(s,...
-
Rank each set of compounds in order of increasing acidity: a. b.
-
Rank each set of compounds in order of increasing boiling points. (a) Triethylamine, di-n-propylamine, n-propyl ether (b) Ethanol, dimethylamine, dimethyl ether (c) Diethylamine, diisopropylamine,...
-
What are the costs of healthcare, where does the money come from, and where is it spent?
-
Discuss the economic and trade importance of the big emerging markets.
-
Which of the following is (Yes) or is not (No) part of the FASB conceptual framework? _____ Objectives of managerial reporting _____ Elements of financial statements _____ Quantitative...
-
Suppose you started up your own landscaping business. A customer paid you $175 in advance to mow his or heV lawn while he or she was on vacation. You performed landscaping services for a local...
-
You are a division manager at Toyota. If your marketing department estimates that the semiannual demand for the Highlander is Q = 100,000 1.25P, what price should you charge in order to maximize...
-
Ausome has been drilling and doing geochemical sampling across its sites in Northern Canada and one site has repeatedly tested positive for the presence of gold. Preliminary estimates suggest that...
-
Use data from Appendix IIB to calculate S rxn for each of the reactions. In each case, try to rationalize the sign of S rxn . Appendix IIB a. CH4(g) + H(g) CH6(g) b. C(s) + HO(g) CO(g) + H(g) c....
-
What is the molar entropy of a pure crystal at 0 K? What is the significance of the answer to this question?
-
What is the difference between auditing standards and auditing procedures?
-
Da Silva, R., & Rahimi, I. (2004). Issues in implementing CRM: a case study. Issues in Informing Science & Information Technology Once you complete the reading of the case, identify the problem of...
-
Summarize the article The Contemporary Social Context of Youth Suicide In most Western societies, suicide is strongly linked to pejorative social meanings. Unlike the grief associated with other...
-
What algorithms are and how they are used by organizations. Additionally, answer the following questions and include citations/ references to the weekly materials. What is the main points of the...
-
A football quarterback is moving straight backwards when he throws a pass to a player 10.4 m straight downfield. The ball is thrown at an angle of 23 degrees relative to the ground and is caught at...
-
If you save $5,000 at the end of every year for 20 years and invest it at 4%, how much will you have in 20 years? How much would you have if you had saved the $5,000 at the beginning of every year?...
-
On January 1, 2017, the Morgantown Company ledger shows Equipment $32,000 and Accumulated DepreciationEquipment $9,000. The depreciation resulted from using the straight-line method with a useful...
-
For each of the following reactions, express the equilibrium constant: a) H20 (I) H2 (g) + 02 (g) Ke = 1.0x107 b) Fe2 (g) 2F (g) Ke= 4.9 x 10-21 c) C (s) + O2 (g) d) H2 (g) + C2H4 (g) C2H6 (g) Ke =...
-
Construct a graph of the angular velocity of a car wheel as a function of time. (a) Assume the wheel starts from rest and moves with a constant (center of mass) velocity. (b) Assume the car starts...
-
Two crates of mass 5.0 kg and 9.0 kg are connected by a rope that runs over a pulley of mass 4.0 kg as shown in Figure P8.57.? (a) Make a sketch showing all the forces on both crates and the pulley.?...
-
Two crates of mass m 1 = 15 kg and m 2 = 25 kg are connected by a cable that is strung over a pulley of mass m pulley = 20 kg as shown in Figure P8.58. There is no friction between crate 1 and the...
-
Fist Fight. Kyle punched Walt in the face in the parking lot of a local bar, breaking Walt's nose. Kyle was charged with aggravated assault. He claims self-defense; Kyle states that Walt was angry...
-
Assume that you have been asked to provide a position statement for your client to attend mediation in a family law case. The information in this case is the other parent who is the petitioner, Ms....
-
Mid-South Auto Leasing leases vehicles to consumers. The attraction to customers is that the company can offer competitive prices due to volume buying. On September 30, 2024, the company leased a...

Study smarter with the SolutionInn App


