The presence of one of the ANSWER NOW! following ions within a compound indicates that a compound
Question:
The presence of one of the ANSWER NOW! following ions within a compound indicates that a compound is soluble with no exceptions. Which ion?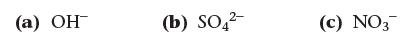
Transcribed Image Text:
(a) OH (b) SO₂²- (c) NO3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (2 reviews)
c Compounds c...View the full answer

Answered By

Marcus Solomon
I am committed to ensuring that my services always meet the clients' expectations.
4.60+
82+ Reviews
117+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
When a pure substance is placed in contact with water, there are three possible outcomes. The substance may do nothing that is, the substance does not dissolve and no visible change takes place. The...
-
A sample containing an alkali sulfate is dried, weighed and dissolved in dilute HCl. Barium chloride solution is added in excess to precipitate barium sulfate, and the precipitate is digested in the...
-
Use the following description of the operations of the RC_Charter2 Company to complete this exercise. ¢ The RC_Charter2 Company operates a fleet of aircraft under the Federal Air Regulations...
-
The top 5 stocks in the S&P 500 index, when ranked by market capitalization, make up 22% of the total market capitalization of the S&P 500 index. Numerical estimates of the mean (or expected) rates...
-
Some businesses choose offshoring to solve the issue of expertise, especially for IT personnel. These companies claim they simply cannot find enough qualified employees in the United States to do...
-
The standard cost card for the single product manufactured by Cutter, Inc., is given below: Manufacturing overhead is applied to production on the basis of standard direct labor-hours. During the...
-
As a gambling facility, MGM Desert Inn, Inc., regularly holds and executes negotiable instruments. During a period of two months, patron William E. Shack Jr. entered MGM and delivered eight checks to...
-
Blair & Rosen, Inc. (B&R) is a brokerage firm that specializes in investment portfolios designed to meet the specific risk tolerances of its clients. A client who contacted B&R this past week has a...
-
Direct Materials Conversion Percent Percent Units Complete Beginning work in process inventory 2,800 100% Complete 25% Units started this period 6,800 Completed and transferred out 7,800 Ending work...
-
What is the net ionic equation for the reaction that occurs when aqueous solutions of KOH and SrCl 2 are mixed? a) K (aq) + CI (aq) KCI(s) b) Sr+ (aq) + 2 OH-(aq) Sr(OH)2 (s) c) H* (aq) + OH (aq) ...
-
Write a chemical formula for each molecular model. a. b. C.
-
For Exercises, find dy for the given values of x and x. y = x 3 - 2x 2 + 3; x = 1, x = -0.1
-
What is referred to as single source? Employing multiple suppliers with the same product Supplied from only one factory A software solution for capacity management The type of capacity model employed
-
Use sales and operations planning to maximize profitability when faced with predictable variability in a supply chain.
-
According to Ferreira and Prokopets, what should a company like Dell consider when evaluating the impact of an offshoring decision?
-
A supply chain strategy involves decisions regarding all of the following except inventory. transportation. new product development. operating facilities.
-
The present value of future cash flow is found by locating the correct factor on a z-table. using a discount factor. plotting the function on a graph. adding the total of all future cash flows.
-
On December 31, 2010, Cochran Company made the following proper year-end adjusting entries: Required 1. Prepare journal entries to record whatever reversing entries you think are appropriate. 2....
-
The area of square PQRS is 100 ft2, and A, B, C, and D are the midpoints of the sides. Find the area of square ABCD. B A
-
Throwing heat. In professional baseball, most pitchers can throw a fastball at a speed of 90 mi/h. For simplicity, assume the pitcher releases the ball just above the center of the pitchers mound....
-
Tom has two ways he could drive home from work. He could take Highway 99 for 45 miles with a speed limit of 65 mi/h, or he could take Interstate 5, which would take him a bit out of the way at 57...
-
According to Newtons third law, for every force there is always a reaction force of equal magnitude and opposite direction. In each of the examples below, identify an actionreaction pair of forces....
-
Green Caterpillar's accumulated owed financial obligations decreased from Year 1 to Year 2. This statement is , because: Accruals actually increased from $0 in Year 1 to $410 million at the end of...
-
To provide incentives, the NWC made the supervisors in Montreal and at the winter trading posts partners. A negative aspect of this organizational form was Group of answer choices the NWC was cash...
-
Arthur Oman, a loan officer at Second National Bank, has been given the unpleasant task of "pulling the plug" on one of the bank's custom- ers, Rebel Discount Drugs. Rebel owes $600,000 on a line of...

Study smarter with the SolutionInn App


