The unit cells for lithium oxide and silver iodide are shown here. Show that the ratio of
Question:
The unit cells for lithium oxide and silver iodide are shown here. Show that the ratio of cations to anions in each unit cell corresponds to the ratio of cations to anions in the formula of each compound.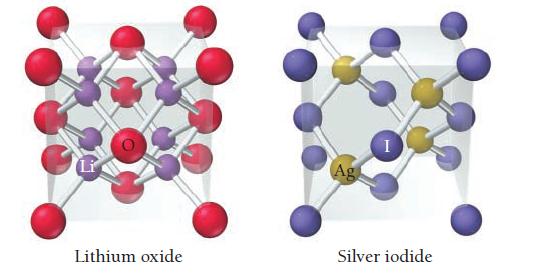
Transcribed Image Text:
Li Lithium oxide Ag Silver iodide
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Lithium oxide Li2O The unit cell for lithium oxide contains four lithium cations Li and four oxide a...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Identify the structure of each of the two unit cells shown in Problem 48 as the rock salt structure, zinc blende structure, fluorite structure, antifluorite structure, or none of these. Problem 48...
-
Identify the structure of each of the two unit cells shown in Problem 47 as the rock salt structure, zinc blende structure, fluorite structure, antifluorite structure, or none of these. Problem 47...
-
The unit cells for cesium chloride and barium chloride are shown here. Show that the ratio of cations to anions in each unit cell corresponds to the ratio of cations to anions in the formula of each...
-
Explain why placing soy sauce in airtight bottles was more successful for long-distance shipping than simply placing the sauce in barrels.
-
On November 1, the Kansas City Post Office Employees Credit Union merged into the Kansas City Telephone Employees Credit Union to form the Communications Credit Union (Credit Union). Systems Design...
-
Distinguish among spoilage, rework, and scrap.
-
The Anchor Glass Container Corporation and its parent company, Consumers Packaging, Inc. (CPI), entered into a series of agreements with Encore Glass, Inc., to supply glass containers of a specific...
-
CVP analysis, income taxes Brooke Motors is a small car dealership. On average, it sells a car for $27,000, which it purchases from the manufacturer for $23,000. Each month, Brooke Motors pays...
-
decribe the health sttsus of unit members (if any of the family describe below)Mother (29yo) and Father (32yo) 2 children (1 elementary, 1 high school) Origin: Mohawk (from Kahnawake) Address: 295,...
-
Consider the rock salt structure in Figure 13.15. What type of structure would result if all the anions were somehow removed, leaving only cations? Sodium chloride (NaCl) CI Na+ A FIGURE 13.15 Sodium...
-
An oxide of rhenium crystallizes with the unit cell shown here (rhenium = gray; oxygen = red). What is the formula of the oxide?
-
Yokam Company is considering two alternative projects. Project 1 requires an initial investment of $400,000 and has a present value of cash flows of $1,100,000. Project 2 requires an initial...
-
Solving Worldly Issues with Entrepreneurship For this project, you will apply your entrepreneurial and enterprising skills to a global issue or problem. Use the following steps to structure your...
-
Who develops Kali Linux? What distribution was the predecessor to Kali Linux? 2. What is the main purpose(s) of the Kali Linux distribution? 3. What are the installation options for Kali Linux?...
-
Have you heard of "The Five Forces model" popularized by Harvard professor Michael Porter, that is a framework for analyzing the attractiveness of an industry? These readings came from the book...
-
How did Cristiano Schena and his team at Caterpillar Brazil use employee engagement and motivation to turn around a struggling business unit? What role did this approach play in sustaining the...
-
GDP and standard of living. What does Gross Domestic Product tell? If GDP increases, what does it mean for the economy? If GDP is higher, does it necessarily mean that living standard of average...
-
Jerry's Ice Cream Parlor is considering a marketing plan to increase sales of ice cream cones. The plan will give customers a free ice cream cone if they buy 10 ice cream cones at regular prices....
-
Apply Jacobis method to the given system. Take the zero vector as the initial approximation and work with four-significant-digit accuracy until two successive iterates agree within 0.001 in each...
-
Using the distribution of particle translational kinetic energy provided in Problem P33.19, derive an expression for the fraction of molecules that have energy greater than some energy . The rate...
-
Imagine a cubic container with sides 1 cm in length that contains 1 atm of Ar at 298 K. How many gaswall collisions are there per second?
-
The vapor pressure of various substances can be determined using effusion. In this process, the material of interest is placed in an oven (referred to as a Knudsen cell) and the mass of material lost...
-
Jennifer, who has a mass of 9 6 kg , has jumped from a plane and is falling through the air at her terminal velocity, a speed of 5 0 m / s . The temperature of the air is 1 5 C . Recall that the...
-
The phase of the satellite is 45d and the phase of the receiver is 165d. The wave length is 500 cm. (a) What is the phase difference? (b) What is the corresponding wavelength for the phase difference
-
a) If the air pressure at sea level is 100.725 kPa and the density of air is 1.25 kg/m3, calculate the thickness of the atmosphere (m) above the earth b) What gauge pressure is experienced by a diver...

Study smarter with the SolutionInn App


