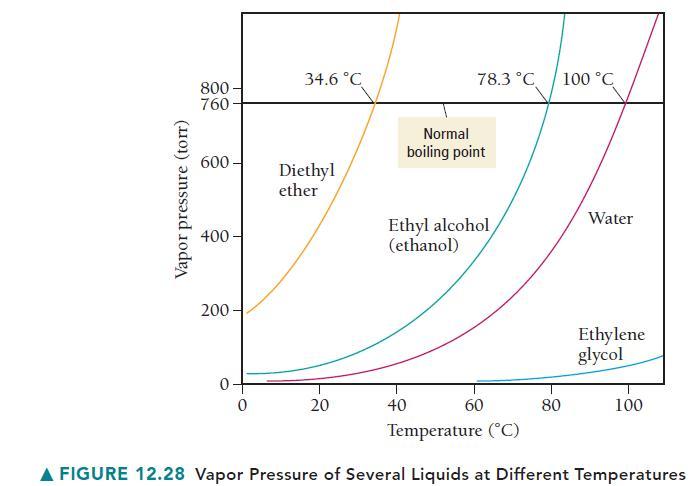
Use Figure 12.28 to estimate the boiling point of water at an external pressure of 200 torr.
Question:
Use Figure 12.28 to estimate the boiling point of water at an external pressure of 200 torr.
(a) 66 °C
(b) 84 °C
(c) 100 °C
(d) 0 °C
Transcribed Image Text:
Vapor pressure (torr) 800- 760 600- 400- 200 0- 0 34.6 °C Diethyl ether 20 78.3 °C 40 Normal boiling point Ethyl alcohol (ethanol) 60 80 100 °C Water Ethylene glycol 100 Temperature (°C) A FIGURE 12.28 Vapor Pressure of Several Liquids at Different Temperatures
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
a According to Figure 1228 water has a vapor pressure of 200 torr at about 6...View the full answer

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each question that uses an interval or ratio scale, include a frequency distribution chart, central tendencies (mean, median, and mode), and dispersion metrics (range, standard deviation ...
-
Determine the dimensions of Nul A, Col A, and Row A for the matrices. A = 0 0 4 7 0 0 0
-
Use Figure 11.7 to estimate the boiling point of carbon tetrachloride, CCl 4, under an external pressure of 250 mmHg. 800 760 700 Chloroform- 600 500 400 Diethyl ether 300 200 Water 100 Carbon...
-
As mentioned in the case, the joint venture between Renault and Mahindra & Mahindra (India) dissolved/failed. Speculate what are the possible socio-cultural issues that triggered this dissolution.
-
Refer to the data for Herald Company in Exercise 15-7. Required: Use the direct method to convert the companys income statement to a cash basis.
-
Find the ABCD parameters for the networks infigure. ZL V2 V1 (a) 12 ZL V1 V2 (b)
-
The Foreign Corrupt Practices act can levy fines of more than five hundred million against companies our size that do not eliminate corruption in their operations abroad. Can you understand why the...
-
At December 31, 2011, the records of NCIS Corporation provided the following selected and incomplete data: Common stock (par $10; no changes during the year). Shares authorized, 200,000. Shares...
-
Inflation Bonds (I Bonds) are safe investments issued by the U.S. Treasury to protect individual's money from loosing value due to inflation. Interest rates on I Bonds are adjusted regularly to keep...
-
Describe the relationship between the state of a substance, its temperature, and the strength of its intermolecular forces.
-
Determine the amount of heat (in kJ) required to vaporize 1.55 kg of water at its boiling point. For water, Hvap = 40.7 kJ/mol (at 100 C). a) 3.50 * 10 3 kJ b) 1.14 * 10 6 kJ c) 2.11 kJ d) 686 kJ
-
Why do jurors often convict on the basis of a false confession even if they think that the confession was probably coerced and could be false?
-
What are two benefits of Conflict Resolution? What are two drawbacks of Conflict Resolution?
-
Define conflict Compare and contrast conflict resolution models Analyze one example of nursing conflict Examine the concept of conflict in nursing work environments Identify sources of generational...
-
Identify all the promotional tools your company the TOMS uses to promote the product ( e . g . , advertising, sales promotion ) and provide current examples for each tool.
-
Identify a conflict resolution strategy and how it applies to each of the conflicts identified. Title this section Conflict Resolution Strategy . For the 2-3 conflicts you describe, address each...
-
Upon completion of this activity, you will be able to: Identify conflict resolution strategies Refer to the conflict resolution strategies discussed by Hynes and respond to the following: Which of...
-
MS Enterprises produces three products X1, X2, and X3 from a joint production process. Data on the process are as follows: Required Determine the value for each lettereditem. Product X1 X2 X3 Total...
-
In Exercises, find the equation of the tangent line at the given point on each curve. 2y 2 - x = 4; (16, 2)
-
A bar having a length of 5 in. and cross-sectional area of 0.7 in. 2 is subjected to an axial force of 8000 lb. If the bar stretches 0.002 in., determine the modulus of elasticity of the material....
-
The rigid pipe is supported by a pin at A and an A-36 steel guy wire BD. If the wire has a diameter of 0.25 in., determine how much it stretches when a load of P = 600 lb acts on the pipe. 4 ft 3 ft...
-
The rigid pipe is supported by a pin at A and an A-36 guy wire BD. If the wire has a diameter of 0.25 in., determine the load P if the end C is displaced 0.075 in. downward. 4 ft 3 ft 3 ft
-
Mariko Koides initial enthusiasm and positive feeling in the new office where she was promoted to soon gave way to despair. The different feeling of the office was complaints between colleagues...
-
Karen just got promoted to general manager of her restaurant, which is part of a chain. Her restaurant is located in Northern British Columbia. When she applied internally for the job, she was told...
-
Venus Corp.s worksheet for calculating current and deferred income taxes for 2006 follows: 2006 2007 2008 Pretax income $1,400 Temporary differences: Depreciation (800) $(1,200) $2,000 ...

Study smarter with the SolutionInn App


