Use line notation to represent each electrochemical cell in Problem 44. Problem 44 Sketch a voltaic cell
Question:
Use line notation to represent each electrochemical cell in Problem 44.
Problem 44
Sketch a voltaic cell for each redox reaction. Label the anode and cathode and indicate the half-reaction that occurs at each electrode and the species present in each solution. Also indicate the direction of electron flow.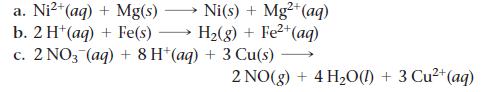
Transcribed Image Text:
2+ Ni(s) + Mg²+ (aq) H₂(g) + Fe²+ (aq) a. Ni²+ (aq) + Mg(s) b. 2 H+ (aq) + Fe(s) c. 2 NO3(aq) + 8 H+ (aq) + 3 Cu(s) 2 NO(g) + 4H₂O(l) + 3 Cu²+ (aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
The line notation for each electrochemical cell is represented as follows a N...View the full answer

Answered By

Ajeet Singh
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions.
I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life.
I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge.
I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields.
Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a teacher. I have done plenty of studying and lots of learning on many exciting and challenging topics. All these experiences have influenced my decision to take on the teaching role in various capacities. As a tutor, I am looking forward to getting to understand your needs and helping you achieve your academic goals. I'm highly flexible and contactable. I am available to work on short notice since I only prefer to work with very small and select groups of students.
4.90+
7+ Reviews
15+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Use line notation to represent each electrochemical cell in Problem 43. Problem 43 Sketch a voltaic cell for each redox reaction. Label the anode and cathode and indicate the half-reaction that...
-
Sketch a voltaic cell for each redox reaction. Label the anode and cathode and indicate the half-reaction that occurs at each electrode and the species present in each solution. Also indicate the...
-
Sketch a voltaic cell for each redox reaction. Label the anode and cathode and indicate the half-reaction that occurs at each electrode and the species present in each solution. Also indicate the...
-
Why must we recognize and address challenges caused by diversity and work to implement a more inclusive healthcare workforce?
-
Summer Diaz has just assumed the duties of controller for Market Research Company. She is concerned that the companys methods of cost planning and control do not accurately track the operations of...
-
Oliva Corporation is a small company that manufactures high-end stereo equipment. Oliva sells its products primarily via specialty audio-video stores. The discerning buyers of the firms products have...
-
On July 31, 2010, the Accounts Receivable balance of Questor Application, Inc., is $320,000. The Allowance for Uncollectible Accounts has a $6,400 credit balance. Questor prepares the following aging...
-
1. Discuss and contrast the features of the retirement plans offered by Analytical Solutions and Precision Manufacturing. 2. Which plan do you think is more desirable? Consider the features,...
-
There are several issues/problems that Greenkeepers have to recognise and address following the construction and Grow-in of new USGA putting greens. Identify the key problems; give your views as to...
-
Make a sketch of the voltaic cell represented by the line notation. Write the overall balanced equation for the reaction and calculate E cell . Sn(s) Sn+ (aq) || NO(g) |NO3(aq), H+ (aq) | Pt(s)
-
Consider the voltaic cell: a. Determine the direction of electron flow and label the anode and the cathode. b. Write a balanced equation for the overall reaction and calculate E cell . c. Label each...
-
Assume that, in 2016, ABB purchased a new automation technology for SFr500 million. They paid this on credit and wont be due to actually pay for the automation technology until 2018. The managers of...
-
Based on the concepts in the Brett, Behfar, and Kern (2006) article, identify and describe a situation where you experienced one of the four barriers. Describe what you did to address the barrier and...
-
What are the advantages/disadvantages of Strategic Alliances?
-
Does the Walmart HR department have a central/key role in creating the workforce?
-
Rearrage the equation to isolate a . ( a + b / c ) ( d - e ) = f
-
Actual and estimated data for manufacturing overhead, direct labor cost, direct labor hours, and machine hours for the most recent fiscal year are summarized here: Direct labor cost Manufacturing...
-
In alphabetical order below are balance sheet items for Mendoza Company at December 31, 2017. Kathy Mendoza is the owner of Mendoza Company. Prepare a balance sheet, following the format of...
-
Southwestern Punch was made by Frutayuda, Inc. and sold in 12-ounce cans to benefit victims of Hurricane Zero. The mean number of ounces placed in a can by an automatic fill pump is 11.7 with a...
-
The electric flux through a closed surface is zero. Must the electric field be zero on that surface? If not, give an example.
-
If the flux of the gravitational field through a closed surface is zero, what can you conclude about the region interior to the surface?
-
Under what conditions can the electric flux through a surface be written as EA,where A is the surface area?
-
Everything Is Not What It Seems You are a perinatal unit coordinator at a large teaching hospital. In addition to your management responsibilities, you have been asked to fill in as a member of the...
-
We met with Christine Smith, a human resources representative, on September 9 th to discuss the city-run residential addiction clinic. She voiced some concerns regarding the nature of staff,...
-
If following code was executed, what is the final value of variable y? y=2; for k=0: 3 y = y +2*k; end

Study smarter with the SolutionInn App


