Use the mass spectrum of europium to determine the atomic mass of europium. Intensity % 100% 50%
Question:
Use the mass spectrum of europium to determine the atomic mass of europium.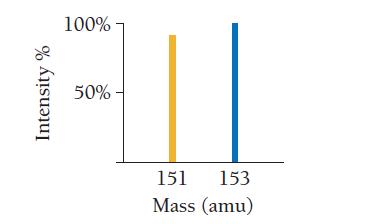
Transcribed Image Text:
Intensity % 100% 50% - 151 153 Mass (amu)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
here is how to determine the atomic mass of europium using its mass s...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Use the mass spectrum of rubidium to determine the atomic mass of rubidium. Intensity % 100% 50%- 85 87 Mass (amu)
-
Mass spectrometry is more often applied to molecules than to atoms. We will see in Chapter 3 that the molecular weight of a molecule is the sum of the atomic weights of the atoms in the molecule. The...
-
The atomic mass of fluorine is 18.998 amu, and its mass spectrum shows a large peak at this mass. The atomic mass of chlorine is 35.45 amu, yet the mass spectrum of chlorine does not show a peak at...
-
On January 1, 20x1, Allan Co. purchased 400,000 bonds for 392,000. The bonds mature on January 1, 20x5 and pay 12% annual interest beginning January 1, 20x2. Transaction costs are negligible. The...
-
Recall Enterprises has no debt. Its current total value is $90 million. Ignoring taxes, what will Recalls value be if it sells $30 million in debt? Suppose now that Recalls tax rate is 40 percent....
-
A thin, homogeneous wire is bent to form the perimeter of the figure indicated. Locate the center of gravity of the wire figure thus formed. r= 150 mm r= 75 mm
-
Tandrin Aviation Holdings Ltd. agreed to sell a jet aircraft to Aero Toy Store, LLC, for \($31.75\) million. ATS paid a \($3\) million deposit to a third party with the balance due upon delivery....
-
Classification IssuesIntangibles) Presented below and on the next page is a list of items that could be included in the intangible assets section of the balance sheet. 1. Investment in a subsidiary...
-
Create this gui on java that supports rational calculations using this code as the logic, make it look like the picture below. Will thumbs up . public class Rational extends Object implements...
-
Suppose youre using depth 0 decision trees, which simply return the weighted majority class of the data points as the classification, as the weak learner. Imagine that, at the first iteration, 80% of...
-
Silicon has three naturally occurring isotopes (Si-28, Si-29, and Si-30). The mass and natural abundance of Si-28 are 27.9769 amu and 92.2%, respectively. The mass and natural abundance of Si-29 are...
-
Write the symbol for each element and classify it as a metal, nonmetal, or metalloid. a. Gold b. Fluorine c. Sodium d. Tin e. Argon
-
The shelf life (in hours) of a certain perishable pack-aged food is a random variable whose probability density function is given by Find the probabilities that one of these packages will have a...
-
One way to measure the strength of a magnetic field is with a flip coil. Suppose a 200 turn, 4.0 cm diameter with a resistance of 2.0 is connected to a 1.0 F capacitor. The coil is held perpendicular...
-
You are expecting a 1 7 % return on the S&P 5 0 0 over the next year. At the same time, you expect the T - bill rate to be 1 % . You have estimated the beta on Macarie Corp to be 1 . 5 . Using CAPM,...
-
If engineering management is broadly defined to include the generalmanagement responsibilities engineers can grow into, how it differs from ordinarymanagement? Using an example that you are aware of,...
-
Define gaap 2. List 8-10 principles and define them
-
Create a Wellness program for the PWC corporation Inc, Ontario, Canada. Explain what a wellness program is, what is the purpose of it, how much does it cost, how does it work, who benefits from it,...
-
One year from today, Daisy Swaine is scheduled to receive a $40,000 payment from a trust fund her father established. She wants to buy a car today but does not have the money. A friend has agreed to...
-
l ask this second time correnct answer is 38,01 can we look pls Consider a non-conducting rod of length 8.8 m having a uniform charge density 4.5 nC/m. Find the electric potential at P, a...
-
Each of the following alcohols can be prepared via reduction of a ketone or aldehyde. In each case, identify the aldehyde or ketone that would be required. a. b. c. d. .
-
Is the ratio of fugacity to pressure greater to or less than one if the attractive part of the interaction potential between gas molecules dominates?
-
Each of the following alcohols can be prepared via reduction of a ketone or aldehyde. In each case, identify the aldehyde or ketone that would be required. a. b. c. d. HO. HO.
-
Let G = (N,A) be an undirected graph. (a) Suppose the arcs of G have with nonnegative weights. Explain how you could modify Dijkstra's algorithm (not G) to find a shortest path from a designated node...
-
Use counting to determine the whole number that corresponds to the cardinality of these sets. (a) C = {xlx = N and (x-8)(x-3)=0} (b) D = {x|x EN, 1x 100 and x is divisible by both 6 and 7} (a) n(C) =...
-
Gradient of the Rayleigh Quotient For a symmetric matrix A and a nonzero vector , the Rayleigh Quotient is defined by xT Ax xTx In the case where is an eigenvector, then the Rayleigh Quotient will...

Study smarter with the SolutionInn App


