Use the mass spectrum of rubidium to determine the atomic mass of rubidium. Intensity % 100% 50%-
Question:
Use the mass spectrum of rubidium to determine the atomic mass of rubidium.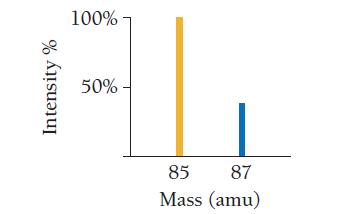
Transcribed Image Text:
Intensity % 100% 50%- 85 87 Mass (amu)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
The image you sent shows the mass spectrum of rubidium The mass spectrum shows two pea...View the full answer

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Mass spectrometry is one of the most versatile and powerful tools in chemical analysis because of its capacity to discriminate between atoms of different masses. When a sample containing a mixture of...
-
Use the mass spectrum of europium to determine the atomic mass of europium. Intensity % 100% 50% - 151 153 Mass (amu)
-
Mass spectrometry is more often applied to molecules than to atoms. We will see in Chapter 3 that the molecular weight of a molecule is the sum of the atomic weights of the atoms in the molecule. The...
-
The e-commerce business in China has entered a golden period, with transaction volume of online trading reaching 21.86 billion yuan (US$2.64 billion) in 2004. With 94 million Internet users, more...
-
Gollum Co. has no debt. Its cost of capital is 9.5 percent. Suppose Gollum coverts to a debt-equity ratio of 1.0. The interest rate on the debt is 7.2 percent. Ignoring taxes, what Gollums new cost...
-
A cubic block of mass m and side length b is placed on a smooth floor. A smooth and rigid rod of length L and with negligible mass is leaning against the block. A sphere of mass M is attached to the...
-
Distinguish between direct and indirect environmental impacts of a division. Provide an example to illustrate the difference.Why is the difference important to a site manager?
-
Using smoothing constants of 0.6 and 0.9 develop forecasts for the sales of Cool-Man air conditioners. Data from Sales of Cool-Man air conditioners have grown steadily during the past 5 years: YEAR...
-
An electron travels at a speed of 4.19107 m/s along the z-axis. What is the magnetic field at the point (0.00 cm,1.00 cm,1.00 cm) when the electron passes through the origin?
-
There is an array A made of N integers. Your task is to choose as many integers from A as possible so that, when they are put in ascending order, all of the differences between all pairs of...
-
How many sulfur atoms are there in 5.52 mol of sulfur?
-
Silicon has three naturally occurring isotopes (Si-28, Si-29, and Si-30). The mass and natural abundance of Si-28 are 27.9769 amu and 92.2%, respectively. The mass and natural abundance of Si-29 are...
-
If the government decided to slow the growth of debt by cutting transfer payments and raising taxes by the same amount, how would this fiscal policy influence the budget deficit and real GDP? CBO...
-
How are team cohesiveness, norms, and performance related?
-
Henry Ford established Ford Motor Company in 1903 in Dearborn, Michigan. At this time, only the rich could afford to purchase vehicles, and Ford wanted to make a vehicle that the common person could...
-
How are norms formed and taught or altered in an organization or group?
-
What are some problems that may crop up in groups and teams?
-
Describe the four types of reinforcement. Which of the four schedules of reinforcement do you think would be most effective at motivating employees to work harder?
-
What are some of the benets of including suppliers in the product development process? Can you think of any risks?
-
A simple random sample of 220 university students were asked what pasta they usually order and with which sauce. The preferences of these respondents are summarised below: Sauce Bolognese Pasta...
-
Predict the major product of the reaction between 1-butanol and: (a) PBr 3 (b) SOCl 2 , py (c) HCl, ZnCl 2 (d) H 2 SO 4 , heat (e) PCC, CH 2 Cl 2 (f ) Na 2 Cr 2 O 7 , H 2 SO 4 , H 2 O (g) Li (h) NaH...
-
What can you conclude about the ratio of fugacity to pressure for N 2 ,H 2 , and NH 3 at 500 bar using the data in Figure 7.10? Figure 7.10 1.5 H2, 1.4 N2 1.3 1.2 1.1 1.0 100 200 300 400 500 600 700...
-
A system consisting of 82.5 g of liquid water at 300. K is heated using an immersion heater at a constant pressure of 1.00 bar. If a current of 1.75 A passes through the 25.0 ohm resistor for 100. s,...
-
Address the situation in the media about the 6 police officers that we're fired and charged for murder for beating Tyre Nichols to death. which aspects of criminal law are present and highlighted by...
-
On August 3, Cinco Construction purchased special-purpose equipment at a cost of $4,700,000. The useful life of the equipment was estimated to be eight years, with an estimated residual value of...
-
Corporate partners Brad and Jen went to the medium sized Bell Law Firm, located in California, to have revocable trusts created for both partners, in 2020. Estate Attorney Al met with both of them,...

Study smarter with the SolutionInn App


