Use the mass spectrum of mercury to estimate the atomic mass of mercury. Estimate the masses and
Question:
Use the mass spectrum of mercury to estimate the atomic mass of mercury. Estimate the masses and percent intensity values from the graph to three significant figures.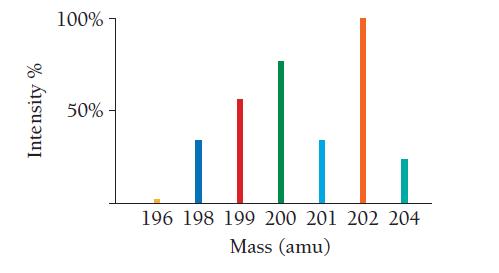
Transcribed Image Text:
Intensity % 100% 50%- 196 198 199 200 201 202 204 Mass (amu)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
The mass spectrum of mercury shows two peaksone at 199 amu and one at 201 amuThe peak at 199 am...View the full answer

Answered By

Chiranjib Thakur
I have no tutoring experience yet, but I can share my skills and knowledge gained from my education and work experiences. I have been a CPA since 2012 with 6 years of work experience in internal auditing and 4 years of work experience in accounting at the supervisory level.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Use the mass spectrum of lead to estimate the atomic mass of lead. Estimate the mass and percent intensity values from the graph to three significant figures. Intensity % 100%- 50%- 204 206 207 208...
-
The masses of the naturally occurring mercury isotopes are 196 Hg, 195.9658 u; 198 Hg, 197.9668 u; 199 Hg, 198.9683 u; 200 Hg, 199.9683 u; 201 Hg, 200.9703 u; 202 Hg, 201.9706 u; and 204 Hg, 203.9735...
-
Mass spectrometry is more often applied to molecules than to atoms. We will see in Chapter 3 that the molecular weight of a molecule is the sum of the atomic weights of the atoms in the molecule. The...
-
Suppose that you just purchased 200 shares of Talk&Tell stock for $60 per share. a. If the initial margin requirement is 71.00%, how much money must you borrow? b. Construct the balance sheet...
-
You have $7,000 to deposit. Regency Bank offers 12 percent per year compounded monthly (1 percent per month), while King Bank offers 12 percent but will only compound annually. How much will your...
-
Financial statement audits: a. Reduce the cost of capital. b. Report on compliance with laws and regulations. c. Assess managements efficiency and effectiveness. d. Overlook information risk. Choose...
-
The phase rule for a system can be written as (a) \(F=C-P-1\) (b) \(F=C-P+1\) (c) \(F=C+P+2\) (d) \(F=C-P+2\) where \(C=\) Number of components \(P=\) Number of phases \(F=\) Degrees of freedom.
-
1. George just hit the jackpot in Las Vegas and won $ 55,000! If he invests it now, at a 12% interest rate, how much will it be worth 15 years from now? 2. Zach would like to have $ 4,000,000 saved...
-
a) What is the future value of $20,000 with an interest rate of 15 percent and one annual period of compounding? b) With an annual interest rate of 15 percent and two semiannual periods of...
-
Using the HCF Balance Sheet on page 226-227 in Table 9A-1, how could you determine the amount of debt principal that will be retired during the next fiscal year? TABLE 9A-1 Harris Memorial Hospital...
-
Fill in the blanks to complete the table. Symbol Z A Si 14 S- 2+ Cu+ 15 32 Number Number Number of p of e of n 14 14 15 34 16 Charge 2- 2+
-
Naturally occurring iodine has an atomic mass of 126.9045 amu. A 12.3849 g sample of iodine is accidentally contaminated with an additional 1.00070 g of 129 I, a synthetic radioisotope of iodine used...
-
A process consists of two steps: (1) One mole of air at T = 800 K and P = 4 bar is cooled at constant volume to T = 350 K. (21 The air is then healed at constant pressure until its temperature...
-
What can be the risk profile on investment by a retired individual, a young professional and a day trader.Describe a long answer for this with good explanation
-
A bond that sells for $899 has a yield of 3.5% and 29 years to maturity. What is the coupon rate? Answer:
-
How does using individual's social media posts constitute discrimination violating EEOC standards? Is there any method that could provide a business a way to use information from an applicant's...
-
June 1.4300 put - premium = 2.01 ($.0201) If spot = $1.4237 and the option expires today, what are your profits?
-
In what ways could the government, educational sector, other private sectors, and non-government organizations help improve other industry (e.g. agriculture) and align the Filipinos' skills and...
-
What is the advantage of a radio media tour (RMT) or satellite media tour (SMT) to the organization and journalists? Are the any disadvantages?
-
Wilsons Auto Repair ended 2011 with Accounts Receivable of $85,000 and a credit balance in Allowance for Uncollectible Accounts balance of $11,000. During 2012, Wilsons Auto Repair had the following...
-
There are five constitutional isomers with molecular formula C 4 H 8 . One of the isomers exhibits a particularly strong signal at M15 in its mass spectrum. Identify this isomer, and explain why the...
-
There are four isomers with molecular formula C 4 H 9 Cl. Only one of these isomers (compound A) has a chirality center. When compound A is treated with sodium ethoxide, three products are formed:...
-
Identify the number of Ï electrons in each of the following compounds. a. b. c. d. e. N'
-
You are a security analyst at a university. The university uses Linux-based tools when systems are discovered to be or suspected to be compromised. Would you use Kali Linux for penetration testing...
-
Like the Windows operating system, Linux also provides keyboard shortcuts for most commands, allowing users to simply press two or more keys on the keyboard at same time instead of typing the exact...
-
Calculate the following. Express your answer as a decimal number with at least four digits past the decimal. (845.6+399.6)(2-3.1)=

Study smarter with the SolutionInn App


