Use the mass spectrum of lead to estimate the atomic mass of lead. Estimate the mass and
Question:
Use the mass spectrum of lead to estimate the atomic mass of lead. Estimate the mass and percent intensity values from the graph to three significant figures.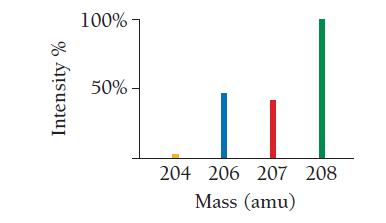
Transcribed Image Text:
Intensity % 100%- 50%- 204 206 207 208 Mass (amu)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
o estimate the atomic mass of lead from the mass spectrum we can take the f...View the full answer

Answered By

Muqadas Javed
I am a mentor by profession since seven years. I have been teaching on online forums and in universities. Teaching is my passion therefore i always try to find simple solution for complicated problems or task grasp them so that students can easily grasp them.I will provide you very detailed and self explanatory answers and that will help you to get good grade. I have two slogans: quality solution and on time delivery.
4.60+
24+ Reviews
144+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Use the mass spectrum of mercury to estimate the atomic mass of mercury. Estimate the masses and percent intensity values from the graph to three significant figures. Intensity % 100% 50%- 196 198...
-
Mass spectrometry is more often applied to molecules than to atoms. We will see in Chapter 3 that the molecular weight of a molecule is the sum of the atomic weights of the atoms in the molecule. The...
-
The mass spectrum of an unknown compound has a molecular ion peak with a relative intensity of 43.27% and an M + 1 peak with a relative intensity of 3.81%. How many carbon atoms are in the compound?
-
Show that = E[(m(X) - X'p)] B = argmind (b) = (E[XX']) E[Xm(X)] = [E[XX'])E[XY]. berk Hint: To show E[Xm(X)] = E[XY] use the law of iterated expectations. then
-
You are scheduled to receive $15,000 in two years. When you receive it, you will invest it for six more years at 8 percent per year. How much will you have in eight years?
-
A financial statement audit: a. Confirms that financial statement assertions are accurate. b. Lends credibility to the financial statements. c. Confirms that financial statements are presented...
-
Gibbs' free energy of a pure fluid approaches at constant temperature (a) Infinity (b) Minus infinity (c) Zero (d) None of these.
-
The following are comparative data for Sunshine State Equipment, Inc., for the 3-year period 20092011. Instructions: 1. From the foregoing data, calculate financial ratios for the three years...
-
Prepare the closing entries and perform the Post-closing trial balance . Trial adjusted balance Account Balance Accounts receivable 119,000 Prepaid insurance 25,000 Materials (supplies) 3,850 Cash...
-
A company's debt is given by a bond that will mature in two years. After two years the company will terminate all activity. The company unlevered equity value in two years can be $17 millions with a...
-
Naturally occurring iodine has an atomic mass of 126.9045 amu. A 12.3849 g sample of iodine is accidentally contaminated with an additional 1.00070 g of 129 I, a synthetic radioisotope of iodine used...
-
An a particle, 4 He 2+ , has a mass of 4.00151 amu. Find the value of its charge-to-mass ratio in C/kg.
-
What was the change in Global Conglomerate's book value of equity from 2014 to 2015 according to Table 2.1? Does this imply that the market price of Global's shares increased in 2015? Explain.
-
A hospitality compOne of the principal and most welcome advantages of a computerized accounting system is its capability to automatically:any is the maker of an $18,000 note to be paid in quarterly...
-
Compare and contrast the gram negative, gram positive, acid fast and, archaea cell walls. with Example
-
Differentiate between plasma membrane of archaea, bacteria and eukaryote structure?
-
Excluding securitization, as it relates to the disintermediation of bank loans, what is securitization? Why would a firm (bank or company) want to securitize an asset? Explain briefly
-
How would you value a company that has been in business for 6-months? You are provided income and balance sheet statements for the firm. Lists the steps that you would follow. How would your...
-
What are the mechanics of organizing a news conference?
-
Select the correct answer for each of the following questions. 1. On December 31, 20X3, Saxe Corporation was merged into Poe Corporation. In the business combination, Poe issued 200,000 shares of its...
-
Malonic acid has two acidic protons: The pKa of the first proton (pK 1 ) is measured to be 2.8, while the pK a of the second proton (pK 2 ) is measured to be 5.7. (a) Explain why the first proton is...
-
Identify a systematic (IUPAC) name for each of the following compounds a. b. c. d. (e) CH 3 (CH 2 ) 4 CO 2 H (f) CH 3 (CH 2 ) 3 COCl (g) CH 3 (CH 2 ) 4 CONH 2 O: NH2
-
Identify the common name for each of the following compounds: a. b. c. d.
-
Discuss the concept of symbiosis in microbial communities, exploring the intricate relationships between different microbial species and their host organisms in various environments .
-
Growth Ltd has decided to expand its business after having an existence for 5 years in retailing. Having established their credit worthiness, they decided to raise funds by issuing bonds. They are...
-
1) An object falls past a window on the fifth floor of a building, when it reaches the bottom of the window it has a speed of 5.45m/s. If the window is 1.5m tall how long does it take for the object...

Study smarter with the SolutionInn App


