What is the net ionic equation for the reaction that occurs when aqueous solutions of KOH and
Question:
What is the net ionic equation for the reaction that occurs when aqueous solutions of KOH and HNO3 are mixed?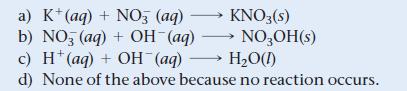
Transcribed Image Text:
a) K+ (aq) + NO3 (aq) b) NO (aq) + OH¯(aq) c) H(aq) + OH(aq) →→→ H₂O(1) d) None of the above because no reaction occurs. KNO3(s) NO3OH(s)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
c H...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
What is the net ionic equation for the reaction that occurs when aqueous solutions of KOH and SrCl 2 are mixed? a) K (aq) + CI (aq) KCI(s) b) Sr+ (aq) + 2 OH-(aq) Sr(OH)2 (s) c) H* (aq) + OH (aq) ...
-
You are developing an accrual-adjusted income statement from a cash income statement. During the year, accounts payable increased. What is the adjustment you need to make?" add ending accounts...
-
What is the net ionic equation for the reaction that occurs when aqueous solutions of KHCO 3 and HBr are mixed? KCH3O (s) a) K+ (aq) + CH3O (aq) b) H+ (aq) + HCO3 (aq) - c) H+ (aq) + OH-(aq) CO(g) +...
-
1. Prepare program using threads in java that can print 10 times the numbers 1,2,3,4,5 in a series. 2. Prepare program using threads and a semaphore in java that can print the numbers 1,2,3,4,5 in a...
-
Figure is a system flowchart for P. Miesing and Companys purchase order event. Prepare a narrative to accompany the flowchart describing this purchase order event. Include in your narrative the...
-
A gas consists of a mixture of He and N2 molecules. Do the lighter He molecules travel faster than the N2 molecules? Explain.
-
Write a program using the programming language of your choice to implement the representation you designed for Review Question 3.3. Have your program solve the problem, and have it show on the screen...
-
On December 31, 2012, Laraboo Corp. issues 11%, 10-year convertible bonds payable with a maturity value of $4,000,000. The semiannual interest dates are June 30 and December 31. The market interest...
-
a) For silicon, if EG decreases by 0.078 eV, by what fraction does n; increase (assume T is constant at 300K)? b) If the temperature rises from 300K to 600K, by what additional fraction does n;...
-
Write complete ionic and net ionic equations for each reaction. (a) 3 SrCl (aq) + 2 Li3PO4(aq) + KOH(aq) (b) HCH3O2(aq) Sr3 (PO4)2(s) +6 LiCl(aq) HO(1) + KCH3O(aq)
-
Write an equation for the precipitation reaction that occurs (if any) when solutions of sodium nitrate and lithium sulfate are mixed.
-
Melbourne Incorporated has 75,000 shares of $2 cumulative preferred shares outstanding as well as 110,000 common shares. There are no dividends in arrears on the preferred shares. The following...
-
Suppose you write a June expiration call option on Tesla with exercise price $85 and at the same time, write a June Tesla put option with exercise price $80. The premium of the call option is $0.90...
-
The following information was taken from the personal records of Justin Knight on May 31, 2021. Opening Balances Cash $3,900 Computer $2,400 House $196,500 Mortgage $103,400 Net Worth $99,400 The...
-
Suppose the Jenson's alpha of a portfolio is 2.5%, while the actual return is 12%. The risk-free rate is 2.5% and the expected return on the market is 12%. What is the Treynor Ratio of the portfolio?
-
Evaluate the thermodynamic properties and behavior of complex fluids, such as liquid crystals, surfactant solutions, and polymeric materials, in terms of phase transitions, self-assembly phenomena,...
-
Interest rate swap Triple A and Triple B have been offered the following rates per annum on a $10 million 5-year loan: Triple A Triple B S&P credit rating AAA BBB Fixed rate 10.5% 13.6% Floating...
-
Identify and explain at least three types of earnings management.
-
1. As a general strategy, would you recommend that Carl take an aggressive approach to capacity expansion or more of a wait-and-see approach? 2. Should Carl go with the option for one facility that...
-
Argon-ion lasers typically generate multi-watt beams in the green or blue regions of the visible spectrum. Determine the frequency of such a 514.5-nm beam.
-
Consider the function where A is a constant. Show that it is a solution of the differential wave equation. Determine the speed of the wave and the direction of propagation. = (1 '2)p (z vt) + 1
-
Show that the function is a solution of the differential wave equation. In what direction does it travel? (y, t) = (y 41)
-
Explain the purpose of database Define what database is Define what database management system is Explain how database applications make databases more useful Describe how data models are used for...
-
3) Let $$ F(x)=\int _{0}^{x} \frac{\ln (1+t)}{t} d t $$ (a) Find the Maclaurin series for F: (b) Use the series in part (a) to evaluate $F(-1)$ exactly and use the result to state its interval of...
-
1. what are the problems the business is facing 2. is it a symptom of something else or a root cause identify it. BELOW IS THE GENERAL INFORMATION ABOUT THE BUSINESS BUSINESS HISTORY riches Kitchen...

Study smarter with the SolutionInn App


