Which combination is the best choice to prepare a buffer with a pH of 9.0? a) NH3;
Question:
Which combination is the best choice to prepare a buffer with a pH of 9.0?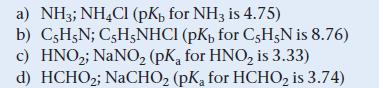
Transcribed Image Text:
a) NH3; NH4Cl (pK, for NH3 is 4.75) b) C5H5N; C5H5NHCl (pK, for C5H5N is 8.76) c) HNO₂; NaNO₂ (pKa for HNO₂ is 3.33) d) HCHO₂; NaCHO₂ (pK, for HCHO₂ is 3.74)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
a NH3...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
2) Give an example for the close loop control system. Explain the working principle of your example and draw its block diagram. (15 Points)
-
Consider the following acids and bases: HCO 2 H K a = 1.8 10 -4 HOBr K a = 2.0 10 -9 (C 2 H 5 ) 2 NH K b = 1.3 10 -3 HONH 2 K b = 1.1 10 -8 Choose substances from the following list that would be...
-
Primo Company, a publicly accountable entity, purchased a property on August 1, 2020 which included land and building for a total price of $3,500,000. Primo financed the purchase of this property by:...
-
Debate on Causes of Unemployment Two economists are debating the cause of a high unemployment rate. One economist argues that there is not enough government spending. The other says high unemployment...
-
Lisah, Inc., manufactures golf clubs in three models. For the year, the Big Bart line has a net loss of $10,000 from sales $200,000, variable costs $180,000, and fixed costs $30,000. If the Big Bart...
-
Wood-Mode Company is involved in the design, manufacture, and installation of various types of wood products for large construction projects. Wood-Mode recently completed a large contract for Stadium...
-
99 percent per month. In 2014, Boling resolved his suit against the gas can manufacturer. Shortly thereafter, Prospect sent Boling a Schedule of Purchases, asserting that Boling owed Prospect...
-
Victory Company uses weighted- average process costing to account for its production costs. Direct labor is added evenly throughout the process. Direct materials are added at the beginning of the...
-
Find all the complex numbers z such that z += 0. Is the set open or closed? {zC: z*+i=0}
-
Two 25.0-mL samples of unknown monoprotic weak acids, A and B, are titrated with 0.100 M NaOH solutions. The titration curve for each acid is shown below. Which of the two weak acid solutions is more...
-
A 50.0-mL sample of 0.200 M sodium hydroxide is titrated with 0.200 M nitric acid. Calculate pH: (a) After adding 30.00 mL of HNO 3 (b) At the equivalence point
-
Construct a finite-state machine for a restricted telephone switching system that implements these rules. Only calls to the telephone numbers 0, 911, and the digit 1 followed by 10-digit telephone...
-
To be negotiable, an instrument must be in writing. (True/False)
-
How would you recommend an American manager plan a negotiation with a Japanese counterpart?
-
American Doll Company begins marketing a new doll with clothes and hair that are highly flammable, and accessories small enough to choke a little child. The Consumer Product Safety Commission can a....
-
Knowing that an instrument has been dishonored puts a holder on notice, and the holder cannot become an HDC. (True/False)
-
A bearer instrument is payable to whoever possesses it. (True/False)
-
You may disagree with the estimates of market size given for the rows in the market-product grid in Figure. Estimate the market size, and give a brief justification for these market segments: (a)...
-
Using the information in P11-2B, compute the overhead controllable variance and the overhead volume variance. Data From Problem 11-2B: Huang Company uses a standard cost accounting system to account...
-
Using Fig. 12.45 , design a problem to help other students better understand wye-delta connected circuits. Solve for the line currents in the Y-Î circuit of Fig. 12.45 . Take Z Î = 60...
-
For the circuit in Fig. 12.43 , determine the current in the neutral line. 880/0 V (+ 25 - j10 2 2 20 2 880-120 V 10 +j5 Q 880/120 V 2
-
Using Fig. 12.41 , design a problem to help other students better understand balanced wye-wye connected circuits. For the Y-Y circuit of Fig. 12.41 , find the line currents, the line\ voltages, and...
-
During Heaton Company's first two years of operations, it reported absorption costing net operating income as follows: Sales (@ $60 per unit) Cost of goods sold (@ $37 per unit) Gross margin Selling...
-
The Physical Therapy Center specializes in helping patients regain motor skills after serious accidents. The center has the following balances on December 31, 2024, before any adjustment: Accounts...
-
November 1 Accepted a $17,000, 180-day, 7% note from Kelly White in granting a time extension on her past-due account receivable. December 31 Adjusted the year-end accounts for the accrued interest...

Study smarter with the SolutionInn App


