Which is the correct Lewis structure for CO 3 2 - ? b) : |:0=C=0: : ::
Question:
Which is the correct Lewis structure for CO32 -?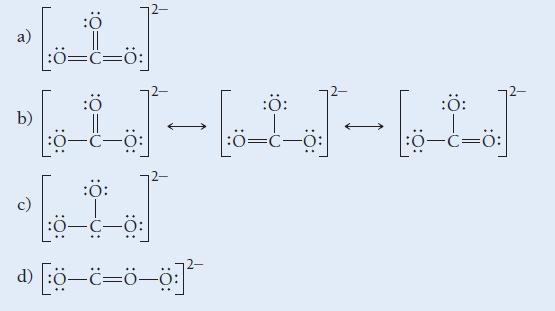
Transcribed Image Text:
ه ده b) :Ö |:0=C=0: :Ö :Ö: - - - :Ö= ھا- ا :Ö: :0–c—0: 20 -=- :Ö: :ة=-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
b ...View the full answer

Answered By

S Mwaura
A quality-driven writer with special technical skills and vast experience in various disciplines. A plagiarism-free paper and impeccable quality content are what I deliver. Timely delivery and originality are guaranteed. Kindly allow me to do any work for you and I guarantee you an A-worthy paper.
4.80+
27+ Reviews
73+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Which is the correct Lewis structure for nitrogen trifluoride? a) :F=N-F: F: c) :F-N-F: :F: b) :F-N-F: F: d) :F-N-F-F:
-
Which is the correct Lewis structure for magnesium bromide? a) 2 Mg+ [Br] c) Mg+2 [Br] b) Mg: Br: d) :Mg: Br:
-
A second Lewis structure can be drawn for one of the nucleophiles in Problem 36. (a) Identify it and draw its alternate structure (which is simply a second resonance form), (b) Is there a second...
-
What is the output of the following program: #include using namespace std; void Push(int x[], int y[], int n) { int i = 0, j = n - 1; while (i
-
Kinkel Corporation makes a product with the following standard costs for direct material and direct labor: Direct material: 1.50 meters at $5.40 per meter . . . . . . . $8.10 Direct labor: 0.25 hours...
-
Describe Finagle a Bagels initial market strategy. What are their options for further growth?
-
For a sample of size n = 15, the following values were obtained: b0 = 3.71, b1 = 8.38, se = 1.13, (x x)2 = 7.71, x = 13.16. Construct a 95% prediction interval for an individual response when x = 8.
-
Following are sales and administrative cost data for Big Jack Burgers for the last four months: Administrative cost is a mixed cost, and sale is a potential cost driver. REQUIRED A. Using the...
-
A three-year bond price is 102.60, the coupon rate is 6% and the bond pays interest annually,theyield-to-maturity is closest to: a. 5.88% b. 5.04% c. 6.26%
-
The reaction between hydrogen and oxygen to form water is highly exothermic. Which statement is true of the energies of the bonds that break and form during the reaction? (a) The energy needed to...
-
Draw the Lewis structure (including resonance structures) for nitromethane (CH 3 NO 2 ). For each resonance structure, assign formal charges to all atoms that have formal charge.
-
Determine the polar moment of inertia of the area shown with respect to (a) Point O, (b) The centroid of the area. 20 in. 20 in.- 12 in. 9 in. 6 in. 12 in. Ellipse
-
Perry Company is in the heating and cooling business. Perry's best-selling item is a comprehensive package for a heating and cooling system maintenance check, tune-up, and energy efficiency test....
-
x1 and x + 2 are both factors of 2x- x + px+q. The value of p is and the value of q is
-
Function g is a transformation of the parent sine function, () = sin(1). 9(x)=sin(2x-5)+1 The phase shift of function g is
-
Provide the calculation for determining the present value of a bond with the following characteristics: a par value of $1,000, a coupon rate of 8.29 percent (paid annually), and a maturity period of...
-
The cash-flow diagram is provided. a. If P = $2,000, A = $250, and i% = 9% per year, then N = ? b. If P = $2,000, A = $250, and N= 8 years, then i = ? c. If A = $250, 1% = 9% per year, and N=4 years,...
-
Assume that your partner and you are in the consumer lending business. A customer, talking with your partner, is discussing the possibility of obtaining a $10,000 loan for three months. The potential...
-
On July 1, 2011, Flashlight Corporation sold equipment it had recently purchased to an unaffiliated company for $480,000. The equipment had a book value on Flashlights books of $390,000 and a...
-
The tank of the air compressor is subjected to an internal pressure of 90 psi. If the inner diameter of the tank is 22 in., and the wall thickness is 0.25 in., determine the stress components acting...
-
The thin-walled cylinder can be supported in one of two ways as shown. Determine the state of stress in the wall of the cylinder for both cases if the piston P causes the internal pressure to be 65...
-
A pressurized spherical tank is made of 0.5-in.-thick steel. If it is subjected to an internal pressure of p = 200 psi, determine its outer radius if the maximum normal stress is not to exceed 15 ksi.
-
A project has annual cash flows of $4,000 for the next 10 years and then $6,500 each year for the following 10 years. The IRR of this 20- year project is 12.41%. If the firm's WACC is 10%, what is...
-
Discuss the role of social identity theory in shaping workplace dynamics. How might these dynamics contribute to intergroup conflict or cohesion within an organization ?
-
Let A(x) = f(t)dt, with f(x) as in figure. 2 1.8- -2 0.5 8 A(x) has a local minimum on (0,6) at x=. A(x) has a local maximum on (0,6) at x=.

Study smarter with the SolutionInn App


