Which reaction occurs at the anode of a lead storage battery? a) Zn(s) + 2 OH-(aq) Zn(OH)2(s)
Question:
Which reaction occurs at the anode of a lead storage battery?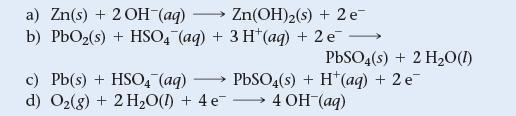
Transcribed Image Text:
a) Zn(s) + 2 OH-(aq) Zn(OH)2(s) + 2 e- b) PbO₂(s) + HSO4 (aq) + 3 H(aq) + 2 e c) Pb(s) + HSO4 (aq) - d) O₂(g) + 2 H₂O(l) + 4 e PbSO4(s) + 2 H₂O(1) PbSO4(s) + H+ (aq) + 2 e¯ →→→→ 4 OH-(aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
What reaction (oxidation or reduction) occurs at the anode of a voltaic cell? What is the sign of the anode? Do electrons flow toward or away from the anode?
-
If a lead storage battery is charged at too high a voltage, gases are produced at each electrode. (It is possible to recharge a lead-storage battery only because of the high overpotential for gas...
-
(a) Briefly explain the difference between oxidation and reduction electrochemical reactions. (b) Which reaction occurs at the anode and which at the cathode?
-
If you wanted to double $1,000 in 10 years' time, what average rate of return would you require on your investment?
-
Using the data in SE 4, SE 5, SE 7, and SE 8, and assuming that Blue Blaze uses the average costing method, assign costs to the units completed and transferred out and to the units in ending...
-
(a) Should you fire Bhandari to reduce operating expenses? (b) If Bhandari is terminated, on what basis could she sue the company? Would she prevail? (c) How could you have structured the...
-
Identify the research scenario, including the general area of focus. Develop a hypothetical research scenario that would necessitate the use of the Action Research Approach and a Posttest With a...
-
Ferraro, Inc. established a stock-appreciation rights (SAR) program on January 1, 2010, which entitles executives to receive cash at the date of exercise for the difference between the market price...
-
Arbon Company has three service departments and two operating departments. Selected data concerning the five departments are presented below: Costs Number of employees Square metres of space occupied...
-
Which of these metals does not act as a sacrificial electrode for iron? (a) Cu (b) Mg (c) Zn (d) Mn
-
Why do some electrochemical cells employ inert electrodes such as platinum?
-
Consider five annual cash flows (the first occurring one year from today): Year Cash Flow 1 .................................. $5 2 .................................. 6 3...
-
Mr. sailor funded the brokerage account with $100,000 and purchased 30 yr. t-strip in march 1987, by end of march, i.r. = 8% Account balance is now about ____ ? In 2006, investor purchased 30yr zero...
-
Jake takes out a $10,000 loan. Later, Jake becomes unable to pay off the loan when the loan balance is $4,000. So, the lender makes a decision to cancel (forgive) the remaining balance on the loan....
-
7. The point (3, 81) is on the graph of the function y = x. a) Find the corresponding coordinates of this point on the graph f(x) = (2x+10)-2. [2K] b) Describe the transformations that must be...
-
Solve the equation. log () x = -3
-
Christophers Custom Cabinet Company uses a job order cost system with overhead applied as a percentage of direct labor costs. Inventory balances at the beginning of 2016 follow: Raw Materials...
-
Identify a problem in our natural world that is the result of overuse or by over mining of one or more natural resources to advance a corporation. Prepare a summary that includes the negative effects...
-
Assume that your audit team has established the following parameters for the examination of ELM's sales transactions: LO G-3 Risk of incorrect acceptance...
-
Collimated red light (656.281 6 nm) from a hydrogen discharge lamp falls perpendicularly onto a transmission grating. The beam emerges forming a red line in the second-order spectrum at an angle of...
-
Light from a laboratory sodium lamp has two strong yellow components at 589.592 3 nm and 588.995 3 nm. How far apart in the first-order spectrum will these two lines be on a screen 1.00 m from a...
-
With Example 10.9 on page 494 in mind, determine the number of grooves a transmission grating must have if it is to resolve the sodium doublet in the first-order spectrum. Compare the results of both...
-
What is version control, and why is it important in collaborative software development? Explain
-
39. Investment A sum of $5000 is invested at an interest rate of 9% per year, compounded continuously. (a) Find the value A(t) of the investment after 7 years. (b) Draw a graph of A(t). (c) Use the...
-
Given the box plot, complete the five-number summary and find the outlier. 25 20 20 15 10 10 5 Ex: 1 Minimum Q1 (first quartile) Median Q3 (third quartile) Maximum Outlier

Study smarter with the SolutionInn App


