Which reaction occurs at the cathode of an electrolytic cell containing a mixture of molten KCl and
Question:
Which reaction occurs at the cathode of an electrolytic cell containing a mixture of molten KCl and ZnCl2?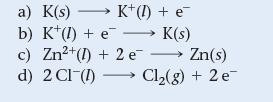
Transcribed Image Text:
a) K(s)→→→→→K+ (1) + e¯ b) K¹ (1) + e K(s) c) Zn²+(1) + 2e →→→ Zn(s) d) 2 CI (1) Cl₂(g) + 2 e-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
c ...View the full answer

Answered By

AJIN kuriakose
I have completed B.Tech in Electrical Engineering & Masters in Power & Control From one of the best universities in India. I got the 99.05 percentile in the Gate Electrical Engineering Exam. I can Help students solving assignments in Electrical subjects like Power Electronics, Control system, Analog, Network Theory & Engineering Mathematics. Clear your fundamentals and develop problem-solving skills and analytical skills to crack the exam.
Get guidance and the opportunity to learn from experienced...
I can provide tuition for Electrical engineering subjects (Power Electronics, Digital electronics, Network Theory, Control System & Engineering Mathematics). The toughest subject of Electrical engineering can be made simple in online classes...
I can also solve it.
1 .I can help you with your assignments or exams or quiz or tutoring.
2. Very strict to the deadlines.
Message me for any help in assignments, live sessions. I am here to help students for all assignments, tests and exams and I will make sure you always get _95% In your subject.
Contact me in solution inn for any help in your semester, projects and for many more things . Also feel free to contact me through solution inn and for any advise related to tutoring and how it works here.thank you.
5.00+
5+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Copper is plated onto the cathode of an electrolytic cell containing CuCl 2 (aq). How long does it take to plate 111 mg of copper with a current of 3.8 A? a) 1.3 x 10 s b) 44 s c) 89 s d) 22 s
-
A constant current of 1.50 amp is passed through an electrolytic cell containing a 0.10 M solution of AgNO3 and a silver anode and a platinum cathode until 2.48 g of silver is deposited. a. How long...
-
A constant current of 1.25 amp is passed through an electrolytic cell containing a 0.050 M solution of CuSO4 and a copper anode and a platinum cathode until 2.20 g of copper is deposited. a. How long...
-
The monochromatic emissivity of a diffuse-surface at 1600 K varies with wavelength in the following manner : = 0.4 for 0
-
McQuary Stone Company produces bricks. Although the company has been in operation for only 12 months, it already enjoys a good reputation. During its first 12 months, it put 600,000 bricks into...
-
Factors, a company in the business of purchasing accounts receivable and lending money to other businesses, asked Unisearch to conduct a Uniform Commercial Code (UCC) search on its behalf for liens...
-
Suppose that in September 2013 a company takes a long position in a contract on May 2014 crude oil futures. It closes out its position in March 2014. The futures price (per barrel) is \($88.30\) when...
-
Jefferson County operates a centralized motor pool to service county vehicles. At the end of 2011, the Motor Pool Internal Service Fund had the following account balances: The following events took...
-
A 2.00 m rod is rotating with an oscillating angular momentum given by sin(2.00) . If a small rocket with oscillating linear momentum 10.0cos(2.00) is fired at =0.00 s , at what time will it need to...
-
How is the cell potential of an electrochemical cell (E cell ) related to the potentials of the half-cells?
-
Is a spontaneous redox reaction obtained by pairing any reduction half-reaction with one listed above it or with one listed below it in Table 20.1? TABLE 20.1 Standard Electrode Potentials at 25 C...
-
A 3.05 m vertical cylindrical exhaust duct from a commercial laundry has an ID of 15.2 cm. Exhaust gases having physical properties approximating those of dry air enter at 316C. The duct is insulated...
-
BBBB Construction Corporation, a domestic corporation, has the following data for 2021 taxable year: Gross income, Phil. P100,000,000 Gross income, USA 50,000,000 Gross income, Japan 50,000,000...
-
Sharon and Gray file a joint tax return and have $750,000 of taxable income in 2021. They had $50,000 of long term capital gains. At what tax rate will they be taxed on their long-term capital gains?...
-
How do advances in single-cell omics technologies, such as single-cell RNA sequencing and single-cell proteomics, enable the elucidation of cellular heterogeneity and lineage trajectories within...
-
Write a function sumArray with: Inputs o the first input is an int array called inputArr the second input is an int that contains the number of elements in inputArr called sizeArr. Returns: sum as...
-
At the close of the month of July, ABC Widgets Incorporated had a balance of $13,498.40. During August, the bank wrote out checks totaling $1,829.35. The company had receivables of $975.46, and its...
-
The economy has seen the unemployment rate increase from 6 percent to 9.5 percent, the inflation rate decrease from 2.8 percent to 1.2 percent, and there has been a 24 percent decline in consumer...
-
What are current assets and current liabilities? How are they different from non-current assets and non-current liabilities?
-
You can make a rough estimate of the thickness of the Earths atmosphere by assuming its density is constant with the value 1.3 kg/m 3 . How tall must the atmosphere be to give the observed pressure...
-
A window in a house is rectangular, with height 40 cm and width 30 cm. The pressure inside the house is normal atmospheric pressure when a hurricane arrives and the wind speed outside is 75 m/s...
-
A backyard waterfall used in landscaping (Fig. P10.94) has water cascading down at a rate of 18 liters/min. (a) If the waterfall is fed using a hose of diameter 2.5 cm, what is the speed of water in...
-
Suppose that the total market value of all final goods and services produced this year in economy X is $4 million. Of the $4 million in goods, $3 million is sold and $1 million is held in inventory....
-
14) Distinguish between Affirmative warranties and promissory warranties as understood in Insurance law. 15) Discuss in detail the claiming of damages as a remedy for breach of contract. 16)...
-
1,000 people are enrolled in a 10-year cohort study. At the start of the study, 225 have diagnosed CVD. Over the course of the study, 80 people who were free of CVD at baseline develop CVD. What is...

Study smarter with the SolutionInn App


