Which transition in the hydrogen atom produces emitted light with the longest wavelength? a. n = 4-
Question:
Which transition in the hydrogen atom produces emitted light with the longest wavelength?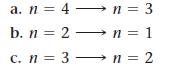
Transcribed Image Text:
a. n = 4- n = 3 b. n = 2n=1 1 c. n = 3n = 2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
The transition in the hydrogen atom that produces emitted light with the longest waveleng...View the full answer

Answered By

Shehar bano
I have collective experience of more than 7 years in education. my area of specialization includes economics, business, marketing and accounting. During my study period I remained engaged with a business school as a visiting faculty member and did a lot of business research. I am also tutoring and mentoring number of international students and professionals online for the last 7 years.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
From the information below, identify element X. a. The wavelength of the radiowaves sent by an FM station broadcasting at 97.1 MHz is 30 million (3.00 107) times greater than the wavelength...
-
Consider a transition of the electron in the hydrogen atom from n = 4 to n = 9. (a) Is E for this process positive or negative? (b) Determine the wavelength of light that is associated with this...
-
Calculate the longest and shortest wavelengths of light emitted by electrons in the hydrogen atom that begin in the n = 6 state and then fall to states with smaller values of n.
-
What is the difference between an optimistic approach and a pessimistic approach to decision making under assumed uncertainty?
-
Selected financial data for Black & Decker and Snap-On Tools for 2007 are presented here (in millions). Instructions (a) For each company, compute the following ratios. (1) Current ratio. (2)...
-
(c) Prove that the lateral displacement d of the emergent beam is given by the relationship where t is the thickness of the plate.(d) A ray of light is incident at an angle of 66.0o on one surface of...
-
Beng-Yu Woo, Xiaoming Li, and Vivian Hsiun created and patented an invention titled Full Duplex Single Chip Video Codec. At the time, Woo, Li, and Hsiun were employees of Infochips Systems, Inc....
-
Janice Sanders, CEO of Pine Crest Medical Clinic, is concerned over the number of times patients must wait more than 30 minutes beyond their scheduled appointments. She asked her assistant to take...
-
A professor designing a class demonstration connects a parallel-plate capacitor to a battery, so that the potential difference between the plates is 255 V. Assume a plate separation of d = 1.72 cm...
-
Determine whether an interference pattern is observed on the other side of the slits in each experiment. a. An electron beam is aimed at two closely spaced slits. The beam produces only one electron...
-
The light emitted from one of the following electronic transitions (n = 4 n = 3 or n = 3 n = 2) in the hydrogen atom causes the photoelectric effect in a particular metal, while light from the...
-
Use z = a + bi to show that z + z = 2a and that z z = 2bi.
-
Use your calculator to evaluate and round the answer to 2 decimal places. 0.03 650 1+ 3.4 0.03 3.4
-
Find the energy usage for traveling a distance of 1 km by an ICE car and a battery electric vehicle considering well - to - wheel energy efficiency. a . Consider that petrol for ICE cars has a...
-
Imagine that you are the financial advisor for a family of five, consisting of a father, a mother, and three young children (ages six, eight, and twelve). Currently, only the mother, Carol, is...
-
Hayes Jewelers is a jewelry wholesaler. Lowell was employed by Hayes as a traveling sales representative. He drove his own vehicle to meet clients and attend trade shows. His practice was to keep the...
-
If you pay $ 2 0 0 0 at the beginning of each year for 1 0 years for your insurance scheme which earns 1 2 % compounded annually, what is the present value of your insurance scheme?
-
Crazy Mountain Sports sells hunting and fishing equipment and provides guided hunting and fishing trips. Crazy Mountain is owned and operated by Karl Young, a well-known sports enthusiast and hunter....
-
Find the work done in pumping all the oil (density S = 50 pounds per cubic foot) over the edge of a cylindrical tank that stands on one of its bases. Assume that the radius of the base is 4 feet, the...
-
Discuss whether the results shown in Figure 17.7 are consistent with local realism. Figure 17.7 300 250 200 150 100 50 -8 -6 -4 -2 4 6. 8. Detector 2 position/mm Coincidence counts
-
An electron and a He atom have the same uncertainty in their speed. What can you say about the relative uncertainty in position for the two particles?
-
Describe the trends in Figure 17.5 that you expect to see as the quantum number n increases. Figure 17.5 n-101 -6 -4 -2 k/(1010 n-15 -1.0 -0.5 0.5 1.0 k(1010 m ) n-5 -0.6 -0.4 -0.2 0.2 0.4 0.6...
-
X is a random variable for a loss. Losses in the year 2000 have a distribution such that E(X^d) -0.025d +1.475d-2.25, d = 10, 11,..., 26 = Losses are uniformly 10% higher in 2001. An insurance policy...
-
In a manufacturing firm, units to be produced are calculated as budgeted sales + desired Blank______. Multiple choice question. work-in-process ending inventory expected beginning work-in-process...
-
A recently married couple hopes to be first-time home buyers in 4 years. Based on their desired neighborhood they estimate they will need $22,000 for a down payment. How much money does the couple...

Study smarter with the SolutionInn App


