Write a ratio showing the relationship between the molar amounts of each element for each compound. a.
Question:
Write a ratio showing the relationship between the molar amounts of each element for each compound.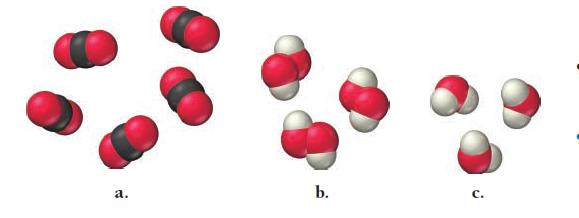
Transcribed Image Text:
a. b.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
a Chemical formula is CO2 and chemical name is carbon dioxide There are one Catom and two ...View the full answer

Answered By

Anurag Agrawal
I am a highly enthusiastic person who likes to explain concepts in simplified language. Be it in my job role as a manager of 4 people or when I used to take classes for specially able kids at our university. I did this continuously for 3 years and my god, that was so fulfilling. Sometimes I've skipped my own classes just to teach these kids and help them get their fair share of opportunities, which they would have missed out on. This was the key driver for me during that time. But since I've joined my job I wasn't able to make time for my passion of teaching due to hectic schedules. But now I've made a commitment to teach for at least an hour a day.
I am highly proficient in school level math and science and reasonably good for college level. In addition to this I am especially interested in courses related to finance and economics. In quest to learn I recently gave the CFA level 1 in Dec 19, hopefully I'll clear it. Finger's crossed :)
4.80+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Write a ratio showing the relationship between the molar amounts of each element for each compound. a. b. J
-
For the Salaries data set, create scatter diagrams showing the relationship between each possible explanatory variable and salaries. Are there any obvious problems? Are there some variables that seem...
-
Write the chemical formula showing the relationship between plants and oxygen, carbon dioxide and sugar. then write the chemical formula showing the relationship between animals and oxygen, carbon...
-
As a preliminary to requesting budget estimates of sales, costs, and expenses for the fiscal year beginning January 1, 2009, the following tentative trial balance as of December 31, 2008, is prepared...
-
Refer back to figure 10.10. What range of returns would you expect to see 68 percent of the time for long-term corporate bonds? What about 95 percent of thetime? Average Standard returndeviation...
-
Consider a \(0.6 \mathrm{~m}\) high and \(2 \mathrm{~m}\) wide double-pane window consisting of two \(4 \mathrm{~mm}\) thick layers of glass \(\left(k=0.80 \mathrm{~W} / \mathrm{m}^{\circ} \mathrm{C}...
-
Imagine that a physician is given the following information about his patient, Mr. Russo: Diagnosis: hypertension. Last blood pressure measurement: 160/100 mmHg. Use this example to discuss the...
-
Developing Standard Costs ColdKing Company is a small producer of fruit-flavored frozen desserts. For many years, Cold- Kings products have had strong regional sales oa the basis of brand...
-
Brittany is 2 7 years old, has one daughter who is blind, and files as Head of Household. What is the Standard deduction?
-
Comprehensive Problem Bug-Off Exterminators December 31, 2019 Unadjusted Bug-Off Exterminators provides pest control services and sells extermination products manufactured by other companies....
-
Determine the number of moles of hydrogen atoms in each sample. a. 0.0885 mol C 4 H 10 b. 1.3 mol CH 4 c. 2.4 mol C 6 H 12 d. 1.87 mol C 8 H 18
-
The American Dental Association recommends that an adult female should consume 3.0 mg of fluoride (F ) per day to prevent tooth decay. If the fluoride is consumed in the form of sodium fluoride...
-
Ed operates a storage business as a sole proprietorship and owns the following assets acquired in 2004: The FMV of the warehouse and the land are \($500,000\) and \($200,000,\) respectively. Ed owns...
-
What practical reason may motivate companies to rarely accrue losses for ongoing litigation?
-
what is the depreciation and CCA amounts for temporary differences in calculating taxable income?
-
How does the interplay of thermodynamic principles and transport phenomena govern the behavior of complex multiphase systems encountered in industrial chemical processes, and how can advanced...
-
Calculate the Electric potential (V) due to an electron at distance 7.5 mm from any source: (Given K=8.99x10 Nm2C-2 and charge of electron is q = 1.6x10-19 C.)
-
An electron is separated from a proton by a distance of 0.53 A. What is the valu of electric field at the location of the electron?
-
The gross national product (GNP) is the sum total of the economic output of the citizens (nationals) of a country. It is an important measure of the wealth of a country. The following table lists the...
-
Test your confidence in the following Project Decisions: SI. # Question 01 02 03 04 05 06 07 08 09 10 How many years did it take to construct the largest Egyptian Pyramid- Pyramid of Cheops? When was...
-
Compound A has molecular formula C 8 H 14 O 2 . Upon treatment with catalytic acid, compound. A is converted into the cyclic hemiacetal. Identify the structure of compound A. , Compound A [H*]
-
Draw a plausible mechanism for each of the following transformations: (a) (b) [TSOH] MENH2 -H20 Et [TSOH] EENH2 -H20
-
Draw a plausible mechanism for each of the following transformations: (a) (b) [TSOH] MENH2 -H20
-
On 1 January 2019 Planet plc began constructing an item of Machinery, the details of this are: Materials 40,000 Labour costs 15,000 Pre-production testing 5,000 Sale of by-products produced as part...
-
The reporting date of Trailer is 31 May 2020. On 1/6/18, Trailer acquired a building at a cost of $90m with a 30 year estimated useful life. On 31/5/19 the building was revalued at $75m. However, the...
-
Calculating a payback period for 5 years based on the below and return on investment. Company initial investment 5.5 Million 2023 As is state: Revenue: $1,896,000; Operating Expenses: $790,260.00...

Study smarter with the SolutionInn App


