Write an equilibrium expression for each chemical equation involving one or more solid or liquid reactants or
Question:
Write an equilibrium expression for each chemical equation involving one or more solid or liquid reactants or products.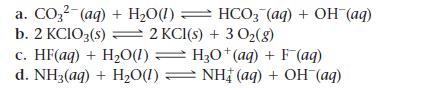
Transcribed Image Text:
a. CO3²- (aq) + H₂O(1) b. 2 KCIO3(s) = 2 KCl(s) + 3 O₂(g) c. HF(aq) + H₂O(1) H3O+ (aq) + F(aq) d. NH3(aq) + H₂O(1) ⇒ NH‡ (aq) + OH (aq) HCO3(aq) + OH (aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a Kc c Ke ...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Write the reaction and the corresponding Kb equilibrium expression for each of the following substances (acting as bases in water). a. NH3 b. CN2 c. Pyridine, C5H5N d. Aniline, C6H5NH2
-
Hydrogen, iodine and hydrogen iodide are in equilibrium in a sealed tube at constant temperature. The equation for the reaction is: H 2 + I 2 2HI(g) H r = 96 kJ mol 1 The partial pressures of each...
-
Sodium carbonate is a diprotic base. Write a chemical equilibrium expression for each of the two successive base reactions with water.
-
As shown in Fig. 4.60, a horizontal beam is hinged to the wall at point A. The length of the beam is 1 = 2 m and it weighs W = 150 N. Point C is the center of gravity of the beam and it is...
-
Payback, Accounting Rate of Return Refer to Exercise 20-5. 1. Compute the payback period for each project. Assume that the manager of the hospital accepts only projects with a payback period of three...
-
Suppose that the disk in Exercise 14.3 rotates at 7200 RPM. a. What is the average rotational latency of this disk drive? b. What seek distance can be covered in the time that you found for part a?
-
Why does application of Lean SE principles to an SDBTF-DPM Engineering Process result in another SDBTF-DPM Engineering Process
-
The manager of a Burger Doodle franchise wants to determine how many sausage biscuits and ham biscuits to prepare each morning for breakfast customers. The two types of biscuits require the following...
-
Pina Colada Corporation manufactures car stereos. It is a division of Berna Motors, which manufactures vehicles. Pina Colada sells car stereos to Berna, as well as to other vehicle manufacturers and...
-
Find and fix the mistake in the equilibrium expression. PC15 (8) PC13(1) + Cl(g) Ke [PC13][C1] [PC]s]
-
Calculate K p for each reaction. a. NO4(8) = 2 NO(g) b. N(g) + 3 H(g) = 2 NH3(g) c. N(g) + O(g) 2 NO(g) K = 5.9 x 10- (at 298 K) K = 3.7 x 108 (at 298 K) K = 4.10 x 10-1 (at 298 K)
-
Shortly after July 31, Morse Corporation received a bank statement containing the following information: July cash transactions and balances on Morses records are shown in the following T-account:...
-
Havana Hats makes the world's best hats. Information for the last eight months follows: Number of Hats Month January Produced 7,800 February 7,250 March 6,150 April 5,250 May 4,500 June 2,100 July...
-
Find at least three potentially problematic data entries (cells) in this table, and decide how you would choose to address them. Would you remove the whole row? Somehow try to guess what the answer...
-
What do you think about this article, down below? do a long paragraph Hijra: India's third gender claims its place in law The history of the hijra community's stretches back toantiquity. But now,...
-
Case: Dodson v. Shrader https://law.justia.com/cases/tennessee/supreme-court/1992/824-s-w-2d-545-2.html Your Role Attorney for Defendant (Shrader) Questions Closing Statement Exhibit Table Answer in...
-
Royal Lawncare Company produces and sells two packaged products-Weedban and Greengrow. Revenue and cost information relating to the products follow: Product Weedban Greengrow Selling price per unit $...
-
Describe important information potentially disclosed in the individual parent and subsidiary companies financial statements that is not found in their consolidated statements. (CFA Adapted)
-
$10,000 was borrowed at 3.5% on July 17. The borrower repaid $5000 on August 12, and $2000 on September 18. What final payment is required on November 12 to fully repay the loan?
-
The current flowing past a point in a device is shown in Fig. 1.25 . Calculate the total charge through the point. i (mA) 10 2 t (ms)
-
Determine the total charge transferred over the time interval of 0 t 10 s when i( t) = 1/2 t A.
-
A total charge of 300 C flows past a given cross section of a conductor in 30 seconds. What is the value of the current?
-
6. Below is a "weird" encoding for a 3-bit wordsize that encodes only 0 and prime values. Suppose we ask a 3-bit computer to add all possible pairs of values and return the result (in the same...
-
Given the two charged particles shown in the figure below, find the electric field at the origin. (Let 91 = -22.00 nC and 92 = 3.00 nC. Express your answer in vector form.) Z = N/C y (cm) 2 92 2 2 -4...
-
Consider the following pseudocode. 1 Algorithm multiSearch( data, target ): 2 data: 3 4 5 6 a list of arrays of integers; in each array the integers are sorted in ascending order%3B the list 'data'...

Study smarter with the SolutionInn App


