Consider the following four titrations (iiv): a. Rank the four titrations in order of increasing pH at
Question:
Consider the following four titrations (i–iv):
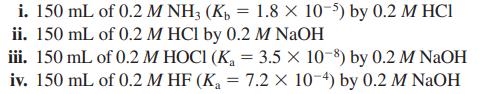
a. Rank the four titrations in order of increasing pH at the halfway point to equivalence (lowest to highest pH).
b. Rank the four titrations in order of increasing pH at the equivalence point.
c. Which titration requires the largest volume of titrant (HCl or NaOH) to reach the equivalence point?
Transcribed Image Text:
i. 150 mL of 0.2 M NH3 (K₂ = 1.8 × 10-5) by 0.2 M HCI ii. 150 mL of 0.2 M HCl by 0.2 M NaOH iii. 150 mL of 0.2 M HOCI (K₁ = 3.5 x 10-8) by 0.2 M NaOH iv. 150 mL of 0.2 MHF (K₁ = 7.2 Xx 10-4) by 0.2 M NaOH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a Rank the four titrations in order of increasing pH at the halfway point to equivalence lowest to h...View the full answer

Answered By

S Mwaura
A quality-driven writer with special technical skills and vast experience in various disciplines. A plagiarism-free paper and impeccable quality content are what I deliver. Timely delivery and originality are guaranteed. Kindly allow me to do any work for you and I guarantee you an A-worthy paper.
4.80+
27+ Reviews
73+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
Consider the following four titrations: i. 100.0 mL of 0.10 M HCl titrated with 0.10 M NaOH ii. 100.0 mL of 0.10 M NaOH titrated with 0.10 M HCl iii. 100.0 mL of 0.10 M CH3NH2 titrated with 0.10 M...
-
Catherine (aged 42) and Johnson (aged 45) have been married for 12 years. Johnson is a project manager of an event company at a monthly salary of $55,000 with an additional one-month salary of...
-
Please write detailed roadmap/solution for all questions below. 1) An output of nmap search is shown below, a) Type the required terminal command and required parameters to obtain the shown output....
-
The following are the Ledger Balance (in thousands) extracted from the books of Vaishnavi Bank Ltd as on March 31, 2016. The bank's Profit and Loss Account for the year ended and Balance Sheet as at...
-
Paul and Julie Leonards two-story home in Pascagoula, Mississippi, is only twelve feet above sea level and less than two hundred yards from the Gulf of Mexico. In 1989, the Leonards bought a...
-
Fill in the Blank. The symmetry of stiffness and flexibility matrices is due to the _________ reciprocity theorem.
-
The 5 - \(\mathrm{kg}\) block is traveling with the speed of \(v=4 \mathrm{~m} / \mathrm{s}\) just before it strikes the 6061-T6 aluminum stepped cylinder. Determine the maximum normal stress...
-
Ballas Co. uses a periodic inventory system. Its records show the following for the month of May, in which 68 units were sold. Instructions Compute the ending inventory at May 31 and cost of goods...
-
Lincoln, a forensic investigator, collected evidence from a crime scene. He used some hardware and software tools to complete the investigation process. Lincoln then created a report and documented...
-
Potholes All along the sides of Route 10, residents displayed signs saying: "County Executive: Fix this Road!" Wanting to minimize the negative publicity associated with this sign campaign, the...
-
To what reaction does the solubility product constant, Ksp, refer? Table 15.1 lists K sp values for several ionic solids. For any of these ionic compounds, you should be able to calculate the...
-
Calculate the value of the equilibrium constant for each of the following reactions in aqueous solution. a. HCHO + OHCHO + HO b. C,H,O, +H*
-
In Exercises 58, the graph of a quadratic function is given. Write the functions equation, selecting from the following options. f(x) = x + 2x + 1 2 h(x) = x - 1 g(x) = x - 2x + 1 j(x) = -x - 1
-
You are creating a Sales order for a customer. The customer wants to observe how prices change as quantities are adjusted. You need to provide an overview of the different price systems available for...
-
Indus Limited produces a product that passes through two processes. The management is worried about the high rate of wastage attributed to workers and has decided to introduce a wage incentive plan...
-
Address the letter to: Mr. Charles Brooks / Oxford Manufacturing Company / 8721 - 104 Avenue, Edmonton, AB T6J 3K4. 4. Include the following additional information as a subject line: Claim No....
-
Instead of paying outrageous fees for insufficient funds (overdraft), you'll be in much a better shape by: Group of answer choices Managing income and expenses carefully. Paying lower interest....
-
If an organization purchased a new imaging system for $100,000 and it estimated that it would keep the system for five years, then sell it for $25,000. How much would the organization record as...
-
Millions of people suffer from migraine headaches. The costs in work days lost, medication, and treatment are measured in the billions of dollars. A study reported in the Journal of the American...
-
Gopher, Inc. developing its upcoming budgeted Costs of Quality (COQ) with the following information: Expense Item Budget Raw Materials Inspection $ 15,000 EPA Fine 200,000 Design Engineering 15,000...
-
The malonic ester synthesis cannot be used to make 2,2-dimethylhexanoic acid. Explain why not.
-
When a malonic ester synthesis is performed using excess base and 1,4-dibromobutane as the alkyl halide, an intramolecular reaction occurs, and the product contains a ring. Draw the product of this...
-
Starting with ethyl acetoacetate and using any other reagents of your choice, propose an efficient synthesis for each of the following compounds. (a) (b) (c) OH
-
On December 3 1 , 2 0 2 5 , American Bank enters into a debt restructuring agreement with Bonita Company, which is now experiencing financial trouble. The bank agrees to restructure a 1 2 % , issued...
-
On January 1 , 2 0 1 9 , Sunland Corporation acquired equipment at a cost of $ 7 8 6 0 0 0 . Sunland adopted the sum - of - the - years - digits method of depreciation for this equipment and had been...
-
John Reardon purchased 100 shares of Tomco Corporation in December 2019, at a total cost of $1,782. He held the shares for 15 months and then sold them, netting $2,535. During the period he held the...

Study smarter with the SolutionInn App


