Calculate the value of the equilibrium constant for each of the following reactions in aqueous solution. a.
Question:
Calculate the value of the equilibrium constant for each of the following reactions in aqueous solution.
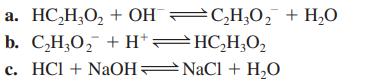
Transcribed Image Text:
a.
HC₂H₂O₂ + OHC₂H₂O₂ + H₂O
b. C,H,O, +H*
a.
HC₂H₂O₂ + OHC₂H₂O₂ + H₂O
b. C,H,O, +H*
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
Image a chemical equation for a chemical reaction HC2H2O2 OH C2H2O2 H2O The equil...View the full answer

Answered By

Cyrus Sandoval
I a web and systems developer with a vast array of knowledge in many different front end and back end languages, responsive frameworks, databases, and best code practices. My objective is simply to be the best web developer that i can be and to contribute to the technology industry all that i know and i can do. My skills include:
- Front end languages: css, HTML, Javascript, XML
- Frameworks: Angular, Jquery, Bootstrap, Jasmine, Mocha
- Back End Languages: Java, Javascript, PHP,kotlin
- Databases: MySQL, PostegreSQL, Mongo, Cassandra
- Tools: Atom, Aptana, Eclipse, Android Studio, Notepad++, Netbeans.
Having a degree in Computer Science enabled me to deeply learn most of the things regarding programming, and i believe that my understanding of problem solving and complex algorithms are also skills that have and will continue to contribute to my overall success as a developer.
I’ve worked on countless freelance projects and have been involved with a handful of notable startups. Also while freelancing I was involved in doing other IT tasks requiring the use of computers from working with data, content creation and transcription.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
Calculate the value for the equilibrium constant for each of the following aqueous reactions. a. NH 3 + H 3 O + NH 4 + H 2 O b. NO 2 - + H 3 O + HNO 2 + H2O c. NH 4 + CH 3 CO 2 - NH3 + CH 3 CO 2 H...
-
a. Calculate the value of Ka for the following acids: i. 0.0200 mol dm 3 2-aminobenzoic acid, which has a pH of 4.30 ii. 0.0500 mol dm 3 propanoic acid, which has a pH of 3.10 iii. 0.100 mol dm 3...
-
A firm is engaged in the production of two types of products. The first product (A) earns a profit of $4 per unit and the second product (B) earns $7 per unit. The sales force requires that at least...
-
A trial balance was extracted from the books of V Baker, and it was found that the debit side exceeded the credit side by 40. This amount was entered in the suspense account. The following errors...
-
Richard Vanderbrooks home in New Orleans, Louisiana, was insured through Unitrin Preferred Insurance Co. His policy excluded coverage for, among other things, flood, surface water, waves, tidal...
-
Fill in the Blank. The determinantal equation, \(\left|[k]-\omega^{2}[m] ight|=0\), is known as the _________ equation.
-
The simply supported W10 \(\times 15\) structural A-36 steel beam is in the horizontal plane and acts as a shock absorber for the 500-lb block which is traveling toward it at \(5 \mathrm{ft} /...
-
Mississippi Company has two decentralized divisions, Illinois and Iowa. Illinois always has purchased certain units from Iowa at $60 per unit. Because Iowa plans to raise the price to $80 per unit,...
-
You will make a web site that consists of 2 different HTML files and 1 CSS file. The files will be named index.html, page2.html, and assign1.css. Please note the case of these files, Index.html is...
-
Review the problem in the Work It Out titled "Interpreting the AD/AS Model." Like the information provided in that feature, Table 24.2 shows information on aggregate supply, aggregate demand, and the...
-
Consider the following four titrations (iiv): a. Rank the four titrations in order of increasing pH at the halfway point to equivalence (lowest to highest pH). b. Rank the four titrations in order of...
-
Consider a solution that contains both C 5 H 5 N and C 5 H 5 NHNO 3 . Calculate the ratio [C 5 H 5 N]/[C 5 H 5 NH + ] if the solution has the following pH values: a. pH = 4.50 b. pH = 5.00 c. pH =...
-
Bill Jovi is reviewing the cash accounting for Nottleman, Inc., a local mailing service. Jovis review will focus on the petty cash account and the bank reconciliation for the month ended May 31,...
-
Which statement is TRUE regarding basis? Basis is: Tied directly to inflation. Never changed after an asset is purchased. The amount of money paid for a piece of property, regardless of other...
-
Your group will conduct a comprehensive financial analysis of a publicly traded corporation and make a presentation to the Board of Directors (professor). Your professor will assign different...
-
Which 3 of the following documents would form part of the reporting process when conducting a routine inspection? Which 3 of the following documents would form part of the reporting process when...
-
For which three reasons should you assign account category and subcategory to a general ledger account? Each correct answer presents part of the solution
-
Suppose you want to purchase a piece of equipment for $10,00 two years from now and another for $35,000 four years from now. How much money should you put into an account at 9% interest compounded...
-
An economist working for a state university wanted to acquire information about salaries in publicly funded and private colleges and universities. She conducted a survey of 623 public- university...
-
An environmentalist wants to determine if the median amount of potassium (mg/L) in rainwater in Lincoln County, Nebraska, is different from that in the rainwater in Clarendon County, South Carolina....
-
The acetoacetic ester synthesis cannot be used to make 3,3-dimethyl-2-hexanone. Explain why not.
-
The product of a Dieckmann cyclization can undergo alkylation, hydrolysis, and decarboxylation. This sequence represents an efficient method for preparing 2-substituted cyclopentanones and...
-
Identify the major product formed when each of the following compounds is treated with Et2CuLi followed by mild acid. (a) (b) (c) CN
-
Please find below table the initial positions of five traders (A, B, C, D, E) on BIST-30 contracts on Day O, together with their only one-way activities during two consecutive days. Please calculate...
-
What were the goals and most important aspects fo the startbuks transformation? elaborate on schultz;s comments aobut redefinding t erole of a forprofit companym, and what you think he means by thi...
-
What are the disadvantages of using an online tax service? Explain.

Study smarter with the SolutionInn App


