For each of the following aqueous reactions, identify the acid, the base, the conjugate base, and the
Question:
For each of the following aqueous reactions, identify the acid, the base, the conjugate base, and the conjugate acid.
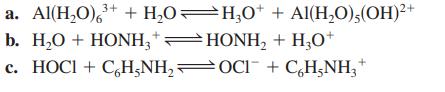
Transcribed Image Text:
3+ a. Al(H₂O) ³+ + H₂O—H₂0* + Al(H₂O),(OH)²+ b. H₂O + HONH3 + HONH₂ + H₂O+ c. HOCI + C₂H5NH₂OCl¯ + C₂H₂NH₂+
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
Answers Equation Acid Base Conjugate Base Conjugate Acid a AlH2O63 H2O AlH2O...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
The conjugate base of diethyl malonate can serve as a nucleophile to attack a wide range of electrophiles. Identify the product that is formed when the conjugate base of diethyl malonate reacts with...
-
Weak base B has a pK b of 6.78 and weak acid HA has a pK a of 5.12. a. Which is the stronger base, B or A ? b. Which is the stronger acid, HA or BH + ? c. Consider the following reaction: B(aq) +...
-
Each of the following reactions has been reported in the chemical literature and gives a single organic product in high yield. Write the structure of the product for each reaction. (a)...
-
A trader depreciates his vehicles at 40% per annum using the reducing balance method, with proportionate depreciation in the year of acquisition. In addition to vehicles acquired on 1 July 2010 for...
-
Rohan, an eighty-three-year-old invalid, employs a nurse, Sarah, to care for him. Prior to Sarahs employment, Rohan executed a will leaving his entire estate to his only living relativehis...
-
True or False. The generalized mass matrix of a multi-degree-of-freedom system is always diagonal.
-
Determine the vertical displacement of point \(C\) of the simply supported 6061-T6 aluminum beam. Consider both shearing and bending strain energy. A 100 kip C La -1.5 ft- -1.5 ft- B
-
Medidata Inc. has identified three risk opportunities for their new medical database project. One is an opportunity to extend the database to include doctors as well as hospitals. This has a...
-
Write two complex numbers z = a + bi and z2 = c + d with a, b, c and d not equal to 0 or 1 with a product of 4+221.
-
Kate, Tina and Naomi - leading Gold coast civil engineering, construction and finance consultants - set up their own firm, KTN Consulting Pty Ltd (KTN). The business expanded rapidly. A joint venture...
-
The pH of a sample of gastric juice in a persons stomach is 2.1. Calculate the pOH, [H + ], and [OH - ] for this sample. Is gastric juice acidic or basic?
-
How many significant figures are there in the following numbers: 10.78, 6.78, 0.78? If these were pH values, to how many significant figures can you express the [H + ]? Explain any discrepancies...
-
How much more massive is an average titanium (Ti) atom than a 12 6 C atom? (Use the periodic table for the atomic mass of titanium.)
-
Total Product (Q) a) Given the following information for Kamaguta Traders. Total Fixed Cost Total Variable Costs (TFC) (TVC) 0 100 0 1 100 90 2 100 170 3 100 240 5 45 100 300 100 370 6 100 450 7 100...
-
a) Given the following information. Construct a graph and derive a market equilibrium. [5 Marks] Thousands of tomatoes Price per Kg demanded (Kg) Thousands of tomatoes supplied (Kg) 85 34 72 80 37 73...
-
a) Compare and contrast the capitalist market system and the command system. [6 Marks] b) Discuss how prices of substitutes and complements, income, cost of doing business can affect supply of flour...
-
a) Distinguish between the following terms: i) Microeconomics and macroeconomics. ii) Positive and normative economics. [2 Marks]. [2 Marks] b) Using a production possibility curve, explain the...
-
a) Using an illustration. Explain the law of demand. [3 Marks] b) Suppose you are a manager of Maneno Traders, explain to the Board of Directors 4 factors that may influence your demand curve for...
-
Refer to Exercise. a. Calculate the correlation between the independent variables. Does it indicate the presence of multicollinearity? Explain. b. Test the correlation between each independent...
-
A report from the college dean indicates that for the previous semester, the grade distribution for the Department of Psychology included 135 As, 158 Bs, 140 Cs, 94 Ds, and 53 Fs. Determine what kind...
-
For each of the following pairs of compounds, identify which compound is the stronger base: (a) (b) (c) (d) N- N- N- N-
-
Rank the following compounds in terms of increasing basicity: N. Br z-
-
When (E)-4-amino-3-buten-2-one is treated with molecular hydrogen in the presence of platinum, the resulting amine is more basic than the reactant. Draw the reactant and the product, and explain why...
-
Two uncharged spheres are separated by 2.60 m. If 2.60 1012 electrons are removed from one sphere and placed on the other, determine the magnitude of the Coulomb force (in N) on one of the spheres,...
-
Jorgansen Lighting, Incorporated, manufactures heavy - duty street lighting systems for municipalities. The company uses variable costing for internal management reports and absorption costing for...
-
Update and Approve A point charge +2Q is at the origin and a point charge -Q is located along the x axis at x = d as in the figure below. Find a symbolic expression for the net force on a third point...

Study smarter with the SolutionInn App


