Two variations of the octahedral geometry (see Table 4.1) are illustrated below. Which of the compounds/ions Br
Question:
Two variations of the octahedral geometry (see Table 4.1) are illustrated below.
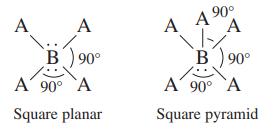
Which of the compounds/ions Br3-, CIF3, XeF4, SF4, PF5, CIF5, and SF6 have these molecular structures?
Transcribed Image Text:
A A B 90° A 90° A Square planar 90° A A ⁹0⁰ A 1/ B) 90° A 90° A Square pyramid
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
The square planar molecular structure is shown on the left in the imageand the square pyramidal molecular structure is shown on the right The followin...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
Write a paper describing a current problem or challenge, how ML or AI could help with this problem, and then describe what this application would be able to do.
-
When a pure substance is placed in contact with water, there are three possible outcomes. The substance may do nothing that is, the substance does not dissolve and no visible change takes place. The...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
It is proposed to use water instead of refrigerant-134a as the working fluid in air-conditioning applications where the minimum temperature never falls below the freezing point. Would you support...
-
Are devaluations of pegged exchange rates totally unexpected?
-
The data in Table 14 represent the two-year average percentage of persons living in poverty, by state, for the years 20122013. Draw a stem-and-leaf plot of the data. Approach Step 1 Treat the integer...
-
What are the different types of schemes associated with complex frauds?
-
Recording Adopted Budget. The City of Marion adopted the following General Fund budget for fiscal year 2011: Estimated revenues: Taxes................$3,000,000 Intergovernmental revenues...........
-
Ho: 5. Now let's consider the effects of comparing responses across samples. Suppose that groups 1 and 2 used the same people, and that responses for individuals 1 to 8 are listed in each row....
-
Indigo is the dye used in coloring blue jeans. The term navy blue is derived from the use of indigo to dye British naval uniforms in the eighteenth century. The structure of the indigo molecule is a....
-
Show how 2s orbitals combine to form bonding and antibonding molecular orbitals. Show how 2p orbitals overlap to form bonding, bonding, antibonding, and antibonding molecular orbitals.
-
Estimate the vapor pressure of ethanol at 37C using thermodynamic data. Express the result in millimeters of mercury.
-
Goodsons has variable costs of $2 per unit and fixed costs of $40,000. Goodsons' selling price is $5 per unit. How much profit will Goodsons earn if it cuts its selling price to $3 per unit, and...
-
The Daniel Insurance Agency reported revenues of $34,000 and expenses of $38,300 for the current period. What was the final figure reported on the company's income statement?
-
PARTICIPATION ACTIVITY 1.4.2: Common syntax errors. Treat the following lines of code as a continuous program. Find and click on the three syntax errors. 1) triangle_base= 0 # Triangle base (cm)...
-
The Financial Post is writing an editorial about recent changes to personal income tax that will affect tax returns that are due within the next few months. By doing this the Post is most likely...
-
Green Landscaping, Inc. is using net present value (NPV) when evaluating projects. Green Landscapings cost of capital is 11.71 percent. What is the NPV of a project if the initial costs are...
-
Why does the purchase of a one-year insurance policy four months ago give rise to insurance expense in the current month?
-
Assume that your audit team has established the following parameters for the examination of ELM's sales transactions: LO G-3 Risk of incorrect acceptance...
-
An unknown salt is either NaCN, NaC 2 H 3 O 2 , NaF, NaCl, or NaOCl. When 0.100 mole of the salt is dissolved in 1.00 L of solution, the pH of the solution is 8.07. What is the identity of the salt?
-
A 0.050-M solution of the salt NaB has a pH of 9.00. Calculate the pH of a 0.010-M solution of HB.
-
Arrange the following 0.10 M solutions in order of most acidic to most basic. KOH, KNO3, KCN, NHCH, HC1
-
What is the kinetic energy of a 126.7 cm thin uniform rod with a mass of 442.2 g that is rotating about its center at 4.82 rad/s? Give your answer in Joules.
-
Your company operates in the chemical industry and is now considering the addition of a new line of detergents. The project costs $ 1 million and is estimated to generate free cash flows of $ 2 5 0 ,...
-
Sadiq Sega raga has recently received a special order from a client to supply 1 0 gates within a month for RM 8 0 , 0 0 0 each. If the company decides to accept this order, following cost information...
Queer Justice The Criminalization Of Lgbt People In The United States - ISBN: 0807051152 - Free Book

Study smarter with the SolutionInn App


