A gas chromatograph is calibrated for the analysis of isooctane (a major gasoline component) using the following
Question:
A gas chromatograph is calibrated for the analysis of isooctane (a major gasoline component) using the following data: 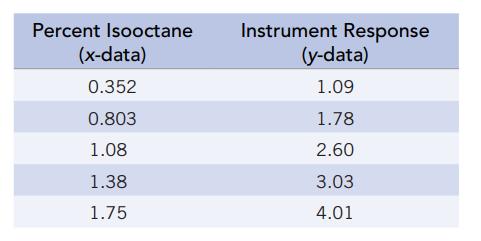
If the instrument response is 2.75, what percentage of isooctane is present?
Transcribed Image Text:
Percent Isooctane (x-data) 0.352 0.803 1.08 1.38 1.75 Instrument Response (y-data) 1.09 1.78 2.60 3.03 4.01
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
To determine the percentage of isooctane x corresponding to an instrument response of 275 y we can u...View the full answer

Answered By

Lilian Nyambura
Hi, am Lilian Nyambura, With extensive experience in the writing industry, I am the best fit for your writing projects. I am currently pursuing a B.A. in Business Administration. With over 5 years of experience, I can comfortably say I am good in article writing, editing and proofreading, academic writing, resumes and cover letters. I have good command over English grammar, English Basic Skills, English Spelling, English Vocabulary, U.S. English Sentence Structure, U.K. or U.S. English Punctuation and other grammar related topics. Let me help you with all your essays, assignments, projects, dissertations, online exams and other related tasks. Quality is my goal.
4.80+
378+ Reviews
750+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
A gas chromatograph is an instrument that measures the amounts of various compounds contained in a sample by separating the various constituents. Because different components are flushed through the...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Federal government budgeting is the forecast or estimate of revenue and expenditures for each fiscal year. The federal budget, like a household budget, itemizes government spending for the following...
-
How does the range of hFE (Fig. 3.23j, normalized from hFE = 100) compare with the range of hfe (Fig. 3.23f) for the range of IC from 0.1 to 10 mA?
-
Anchor.com, a computer consulting firm, has decided to write off the $7,130 balance of an account owed by a customer. Journalize the entry to record the write-off, (a) Assuming that the allowance...
-
A binary symmetric channel has probability p = 0.05 of incorrect transmission. If the code word c = 011011101 is transmitted, what is the probability that (a) We receive r = 011111101? (b) We receive...
-
Derive the kinematic and dynamic conditions needed in the analysis. Set up the equations to find the constants. Now require that the determinant of the coefficient matrix should be zero to obtain a...
-
The following table shows how average share prices jump (in percentage) after the announcement that the stocks will be cross-listed (see Miller, 2000). The price response should be interpreted as...
-
system Solve the problem using two variables and a system of two equations. Solve the system by the method of your choice. Note that the problem could be a dependent or inconsistent A civil engineer...
-
A general chemistry class carried out an experiment to determine the percentage (by mass) of acetic acid in vinegar. Ten students reported the following values: 5.22%, 5.28%, 5.22%, 5.30%, 5.19%,...
-
You can analyze for a copper compound in water using an instrument called a spectrophotometer. [A spectrophotometer is a scientific instrument that measures the amount of light (of a given...
-
Most genes encode polypeptides, which are functional units of proteins. Explain how the structure and function of proteins produce an organisms traits.
-
Use the British method to design a GP concrete mix that is required to have a specified mean strength of 35 MPa at 28 days. The presence of reinforcement requires a slump of 75 mm and a maximum size...
-
Feldstein tells DiMaria thatGM forecast operating exposure for all its regions on a monthly basis. Then, the "riskiness" of these exposures is calculated by multiplying the net (regional) exposure by...
-
You are required, as the broker to call your client, Wes/Wendy Evans (you/your volunteer may choose the name they prefer to use) and provide them with the outcome of their loan application. Wes/Wendy...
-
A35000$ machine has 6 yeas depreciable life and a 10000$ salvage value at the end of the years. Compute the depreciation schedule and the book value at the end of each year using sum of year's digits...
-
1. The company stock name is Amazon 2.=1.15 52-week change for the stock=27.67% the S&P 500=36.35% the 6-month rate=0.06% The formula for the CAPM is expressed as: R STOCK = R RF + ( R M - R RF )...
-
The following data are from the 20X1 income statement of the Atiyeh Rug Emporium ($ in thousands): The ending inventory was overstated by $20,000 because of errors in the physical count. The income...
-
In a paragraph of approximately 150-200 words, analyze a film or TV/Streaming Show poster of your choosing by focusing on the ways in which representations in the poster are gendered. Include an...
-
Identify the reagents that you would use to accomplish each of the following transformations: a. b. c. d. e. f. -Br OH
-
When the following ester is treated with lithium iodide in DMF, a carboxylate ion is obtained: (a) Draw the mechanism of this reaction. (b) When the methyl ester is used as the substrate, the...
-
Suggest an efficient synthesis for the following transformation: En Br
-
Use the following selected data from Business Solutions's income statement for the three months ended March 31, 2022, and from its March 31, 2022, balance sheet to complete the requirements. Computer...
-
The following information is for the Jeffries Corporation: Product A: Selling price per unit $17.00 Variable cost per unit $12.00 Product B: Selling price per unit $42.00 Variable cost per unit...
-
As the owner of a newly established business specializing in manufacturing and sales of electronic gadgets, you are responsible for overseeing the financial aspects of your business. This includes...

Study smarter with the SolutionInn App


