Balance equations for these reactions that occur in aqueous solution, and then classify each as a precipitation,
Question:
Balance equations for these reactions that occur in aqueous solution, and then classify each as a precipitation, acid–base, or gas-forming reaction. Show states for the products (s, ℓ, g, aq), give their names, and write the net ionic equation.
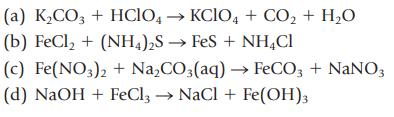
Transcribed Image Text:
(a) K₂CO3 + HCIO4 →KCIO4 + CO₂ + H₂O (b) FeCl, + (NH4)2S → FeS + NHẠCH (c) Fe(NO3)2 + Na₂CO3(aq) → FeCO3 + NaNO3 (d) NaOH + FeCl3 → NaCl + Fe(OH)3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
a KCO HClO KClO CO HO First balance the equation 2 KCO 2 HClO 2 KClO 2 CO 2 HO Now lets classify it ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Write balanced net ionic equations for the reactions that occur in each of the following cases. Identify the spectator ion or ions in each reaction. (a) Cr2 (SO4)3(aq) + (NH4)2CO3(aq) (b) Ba...
-
The converter loop is a net generator of energy (why?) and proper utilization of that energy improves process economics. Assuming that the makeup gas is at 100C and that the purge gas and crude...
-
Balance these redox reactions that occur in aqueous solution. Use whatever water-derived species is necessary; there may be more than one correct balanced equation. a. CrO3 + Ni2+ ( Cr3+ + Ni3+ b....
-
Why would an organization use outplacement strategies before downsizing? Outplacing employees helps former employees develop new skill sets. Outplacement strategies are a means of eliminating problem...
-
Can you suggest an event that would cause a countrys nominal interest rate to rise and its currency to appreciate simultaneously, in a world of perfectly flexible prices?
-
A frequency distribution for the class level of students in Professor Weiss's introductory statistics course is as follows. Class __________________ Frequency Freshman ........................... 6...
-
A four cylinder two stroke petrol engine with stroke to bore ratio of 1.2. develops \(32 \mathrm{~kW}\) bp at 2500 r.p.m. The mean effective pressure on each piston is 8 bar and the mechanical...
-
Coffey Corporation purchased debt investments for $52,000 on January 1, 2010. On July 1, 2010, Coffey received cash interest of $2,340. Journalize the purchase and the receipt of interest. Assume...
-
The Samsung Global Code of Conduct and Business Conduct Guidelines, which direct every one of their representatives, drive their efforts to deliberately oversee consistency and moral dangers. It also...
-
Identify the primary species (atoms, molecules, or ions) present in an aqueous solution of each of the following compounds. Decide which species are Brnsted acids or bases and whether they are strong...
-
Aqueous solutions of ammonium sulfide, (NH 4 ) 2 S, and Hg(NO 3 ) 2 react to produce HgS and NH 4 NO 3 . (a) Write the overall, balanced equation for the reaction. Indicate the state (s, aq) for each...
-
Professor Mason suggests that we modify ANY-SEGMENTS-INTERSECT so that instead of returning upon finding an intersection, it prints the segments that intersect and continues on to the next iteration...
-
What are the key components of a knowledge management framework designed to facilitate tacit knowledge transfer within a complex organizational ecosystem ?
-
Metropolis Health System (MHS) has received a wellness grant from the charitable arm of an area electronics company. The grant will run for 24 months, beginning at the first of the next fiscal year....
-
A thin coil at the origin of the coordinate system has 1 0 0 turns, radius 1 . 4 cm and current 2 A . the magnetic field due to the coil at points on a circle in the xy plane with radius...
-
Murphee Manufacturing is considering purchasing a machine with an expected life of 8 years for $68,000. The machine has an estimated salvage value of $4,000, Murphee expects the new machine to...
-
Assuming the market begins with a 50/50 split of lemon or inferior product and plum or better products. How does a "lemon" push out a "plum" from the market?
-
Refer to the preceding facts for Packards acquisition of Stude common stock. On January 1, 2012, Packard held merchandise acquired from Stude for $10,000. This beginning inventory had an applicable...
-
What impact has the Internet had on the globalization of small firms? How do you think small companies will use the Internet for business in the future?
-
A carbocation is resonance stabilized when it is adjacent to an oxygen atom: Such a carbocation is even more stable than a tertiary carbocation. Using this information, propose a mechanism for the...
-
One liter of fully oxygenated blood can carry 0.18 liters of O 2 measured at T = 298 K and P = 1.00 atm. Calculate the number of moles of O 2 carried per liter of blood. Hemoglobin, the oxygen...
-
Determine whether the pinacol rearrangement, shown in the previous problem, is a reduction, an oxidation, or neither. Resonance stabilized H,SO, eat
-
1- Describe the error in the following code excerpt. for (int i = 1; i = 10; i++) System.out.println("This is the loop body"); 2- Change the code such that when it runs, the print statement is...
-
1. The following matrix gives a set of cognate words meaning 'ox' in several dif- ferent languages. Language Word Pronunciation Avestan uxshan uksan Chuvash vgr English OX oks Finnish hrk hrk...
-
2. Let P be the set of all people. Consider the following predicates, all with domain P: A(x)="x is an athlete" T(x)="x is a teacher" S(x, y)="r is shorter than y" L(x, y)="r likes y" Write the...

Study smarter with the SolutionInn App


