Balance the following equations, and name each reactant and product: Data given in Example 3.1 (a) SF4(g)
Question:
Balance the following equations, and name each reactant and product: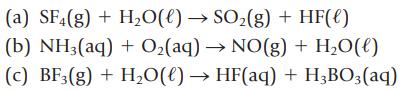
Data given in Example 3.1

Transcribed Image Text:
(a) SF4(g) + H₂O(l) → SO₂(g) + HF(e) (b) NH3(aq) + O₂(aq) → NO(g) + H₂O(l) (c) BF3(g) + H₂O(l) → HF(aq) + H₂BO3(aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
a SFg 2HOl SOg 4HFl Reactants Sulfur tetrafluoride gas and water Products ...View the full answer

Answered By

Amit Choudhary
I'm new in this profession regarding online teaching but previously i used to teach students near my college. I am teaching on online platform since last year and got good support from the students. I'm teaching on platforms like chegg and vedantu and also at my home in free time.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Balance the following equations, and name each reactant and product: Data given in Example 3.1 (a) FeO3(s) + Mg(s) MgO(s) + Fe(s) (b) AlCl3(s) + NaOH(aq) AI(OH)3 (s) + NaCl(aq) (c) NaNO3(s) +...
-
Balance the following equations and write the corresponding ionic and net ionic equations (if appropriate): (a) (b) (c) Ba(OH)-(aq) + HPO 4 (aq )- HCIO4 (aq) + Mg(OH )2 (s)
-
Balance the following equations and write the corresponding ionic and net ionic equations (if appropriate): (a) (b) (c) CH3COOH (aq) + KOH(aq)- .co.(aq) + NaO H (aq) - HNO3(aq ) + Ba(OH)2(aq )-
-
Which statement about the pass-by-reference is NOT true? a.Every time you pass a reference variable to a method, you also pass the object referred by the refenerece variable to the method. b.In the...
-
Describe the factors to consider before developing an aggregate plan.
-
Bill and Kate Theil are not only husband and wife but entrepreneurs who have established three successful businesses. The proposed plan for their latest effort involves a series of international...
-
Records show that the probability is 0.00008 that a truck will have an accident on a certain highway. Use the formula for the Poisson distribution to approximate the probability that at least 5 of...
-
Sunco processes oil into aviation fuel and heating oil. It costs $40 to purchase each 1000 barrels of oil, which is then distilled and yields 500 barrels of aviation fuel and 500 barrels of heating...
-
Why is Microsoft Corp bond a good investment? The Bond has a maturity date of 6/1/2039 and a coupon rate of 5.2000% and is At the current price of 114.234 USD, this equates to a 4.16 percent annual...
-
Write balanced chemical equations for the following reactions. (a) The reaction of aluminum and iron(III) oxide to form iron and aluminum oxide (known as the thermite reaction). (b) The reaction of...
-
Balance the following equations: Data given in Example 3.1 (a) Cr(s) + Cl(g) CrCl3(s) (b) SiO (s) + C(s) Si(s) + CO(g) (c) Fe(s) + HO(g) Fe3O4(s) + H(g)
-
What kind of writing is required for a negotiable instrument?
-
6 10 points eBook Hint The following monthly data are taken from Ramirez Company at July 31: Sales salaries, $600,000; Office salaries, $120,000; Federal income taxes withheld, $180,000; State income...
-
Given the probability density function 10 f(x)= 243 0 Find - E[x] for r21. x(3-x)4 for 0
-
The following is the Excel Solver output of Linear Programming Problem. Objective Allowable Reduced Cost Coefficient Increase 1800000 120000 1600000 1280000 Name Variable-S Variable- M Constraints:...
-
Simplify: x(x+x)
-
Using the trial balance below, prepare the following: -Statement of Comprehensive Income (2 Statement Approach) -Statement of Stockholders Equity -Statement of Financial Position Calculate the...
-
For each of the following research projects, list at least one area or cluster that could be used in obtaining the sample. a. A study of road conditions in the state of Missouri b. A study of U.S....
-
For Problem estimate the change in y for the given change in x. y = f(x), f'(12) = 30, x increases from 12 to 12.2
-
Draw all significant resonance structures for each of the following compounds: a. b. c. d. e. f. g. h. i. j. k. l. . z: N.
-
A sealed flask with a capacity of 1.22 dm 3 contains 4.50 g of carbon dioxide. The flask is so weak that it will burst if the pressure exceeds 9.500 10 5 Pa. At what temperature will the pressure of...
-
A balloon filled with 11.50 L of Ar at 18.7C and one atm rises to a height in the atmosphere where the pressure is 207 Torr and the temperature is 32.4C. What is the final volume of the balloon?...
-
As flextime, consulting, telecommuting, and downsizing make it more difficult for people to donate blood at the workplace, Canadian Blood Services has launched a CRM marketing campaign in Toronto to...
-
Product : my product is portable and affordable 3D printer that uses sustainable materials, such as recycled plastics or plant-based fibers. This product is important because it democratizes access...
-
Direct Method, Reciprocal Method, Overhead Rates Macalister Corporation is developing departmental overhead rates based on direct labor hours for its two production departments-Molding and Assembly....

Study smarter with the SolutionInn App


