Calculate the pH of a 0.12 M aqueous solution of the base aniline, C 6 H 5
Question:
Calculate the pH of a 0.12 M aqueous solution of the base aniline, C6H5NH2 (Kb = 4.0 × 10−10).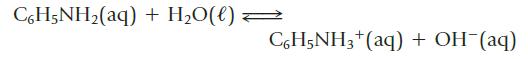
Transcribed Image Text:
CoH5NHz(aq) +H,O(l)< C6H5NH3+ (aq) + OH-(aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (6 reviews)
To calculate the pH of a 012 M aqueous solution of the base aniline C6H5NH2 you can use the concept ...View the full answer

Answered By

Michael Owens
I am a competent Software Engineer with sufficient experience in web applications development using the following programming languages:-
HTML5, CSS3, PHP, JAVASCRIPT, TYPESCRIPT AND SQL.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Lets take a look at the extensive career of Helen Frankenthaler (1928-2011), who found a way to incorporate all of the innovative techniques artists were experimenting with starting in 1945. Her...
-
Calculate the pH of a 0.15 M aqueous solution of aluminum chloride, AlCl3. The acid ionization of hydrated aluminum ion is and Ka is 1.4 10-5.
-
Calculate the pH of a 0.15 M aqueous solution of zinc chloride, ZnCl2. The acid ionization of hydrated zinc ion is and Ka is 2.5 10-10.
-
Sketch the six graphs of the x- and y-components of position, velocity, and acceleration versus time for projectile motion with x 0 = y 0 = 0 and 0 < 0 < 90.
-
What are the advantages and disadvantages of having an employer draw attention to situations where employees use some of their work time to do non-job-related activities? Does labeling this employee...
-
What is a species?
-
At the Diabetes Institute, patients are classified as compliant (they take their medication as they should) or not (they don't). A new program manager has a new idea: Have some patients continuously...
-
The following balances are from the accounts of Hill Components: Direct materials used during the year amount to $59,800, and the cost of goods sold for the year was $68,900. Required Find the...
-
The Gibb's Free Energy (AG) can be used to determine if a reaction is spontaneous as written. It is calculated from two factors, enthalpy and entropy. A. (6) Briefly describe what enthalpy and...
-
Suppose you have $5,000 in savings when the price level index is at 100. (a) If inflation pushes the price level up by 10 percent, what will be the real value of your savings? (b) What is the real...
-
Hemoglobin (Hb) can form a complex with both O 2 and CO. For the reaction at body temperature, K is about 200. If the ratio [HbCO]/[HbO 2 ] comes close to 1, death is probable. What partial pressure...
-
Is it true that in a short-run production process, the marginal cost curve eventually slopes upward because firms have to pay workers a higher wage rate as they produce more output? Explain your...
-
On 21 March 2022, Penny is made redundant by her employer. She receives statutory redundancy pay of 4,750 and an ex gratia compensation payment of 32,000 (none of which ranks as a PILON). Her only...
-
Under the current year basis, for which tax years would the following accounting years form the basis period? (a) year to 31 October 2021 (b) year to 31 March 2022 (c) year to 30 April 2023 (d) year...
-
Throughout 2021-22, Niall is provided by his employer with a diesel-engined motor car which had a list price when new of 24,200. The car was registered in January 2021 and has an emission rating of...
-
Which of the following forms of income from employment would be exempt from income tax in tax year 2021-22? (a) Free meals in the company canteen. (b) Removal expenses of 4,500. (c) 1,000 given to an...
-
Malcolm's gross annual salary is 57,500. He is also entitled to an annual bonus which is based on his employer's profits for the previous calendar year. His bonus for the year to 31 December 2020 was...
-
Jellystone National Park is located 10 minutes away from city A and 20 minutes away from city B. Cities A and B have 200,000 inhabitants each, and residents in both cities have the same income and...
-
Find the work done in pumping all the oil (density S = 50 pounds per cubic foot) over the edge of a cylindrical tank that stands on one of its bases. Assume that the radius of the base is 4 feet, the...
-
Determine the support reactions at and . Take E = 200 MPa, I = 300(10 6 ) mm 4 , A = 21(10 3 ) mm 2 for each member. - 5 m 300 kN m, 3 9. (2, 4 m 2
-
Determine the structure stiffness matrix K for the frame. Take E = GPa, I = 350(10 6 ) mm 4 , A = 15(10 3 ) mm 2 for each member. Joints at and are pins. 2 60 kN 3 |1 2) 2 m - - 2 m 2 4 m
-
Determine the support reactions at pins and . Take E = 200 GPa, I = 350(10 6 ) mm 4 , A = 15(10 3 ) mm 2 for each member. 2 60 kN 3 - 2 m 2 m 4 m 3) 9
-
Derive the weakest precondition for the sequence of assignment statements and their postconditions below: a) x=3y+ 1; y=a-5 {y <1} b) c 3 (1 b+c); b=2*c-1 {b>4}
-
As the world population is rising, speed of transportation is expanding, and new markets are opening, the potential for economic returns (and risks) is rising. Countries integrate economically for...
-
Write a general Matlab code to convert X to the new base (B). Test your code with the following numbers: (2146.527) to (........ (4521.124) to (.............) Enter X=XXXX.XX.X... Old base -b & New...

Study smarter with the SolutionInn App


