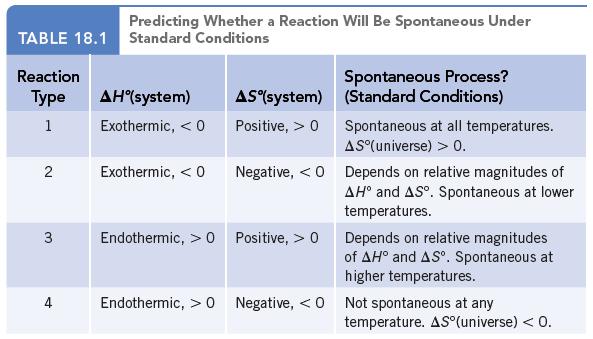
Classify each of the reactions according to one of the four reaction types summarized in Table 18.1.
Question:
Classify each of the reactions according to one of the four reaction types summarized in Table 18.1.![]()
Δr H° = −673 kJ/mol-rxn
Δr S° = 60.4 J/K · mol-rxn![]()
Δr H° = 490.7 kJ/mol-rxn
Δr S° = 197.9 J/K · mol-rxn
Data given in Table 18.1
Transcribed Image Text:
(a) C6H12O6(s) + 6 O₂(g) →6 CO₂(g) + 6 H₂O(l)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
a Since this reaction has enthalpy change negative and entropy change positive Th...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Classify each of the reactions according to one of the four reaction types summarized in Table 18.1. r H = 851.5 kJ/mol-rxn r S = 375.2 J/K mol-rxn r H = 66.2 kJ/mol-rxn r S = 121.6 J/K mol-rxn...
-
Classify each of the following reactions as one of the four possible types summarized in Table 19.3: (a) (b) (c) N2(g) 3 F2(g)2NF3(g) AH249 kJ; AS278 J/K N2(g) + 3C12(g) --> 2NC3(g) AH 460 kJ; AS...
-
Classify each of the following qualitative variables as ordinal or nominative. Explain your answers. Qualitative Variable Categories Personal computer operating system.. Windows XP Windows Vista...
-
When would you suggest that an organization engage in workflow analysis?
-
Despite obtaining a distinction for the project, the supervisor commented that Chris should have discussed the limitations of his data sources more thoroughly. Visit the IPOs website [www.wipo.org]...
-
There is no external input in the circuit in Figure P10\(4 \underline{8}\). (a) Find the zero-input node voltages \(v_{\mathrm{A}}(t)\) and \(v_{\mathrm{B}}(t)\), and the voltage across the capacitor...
-
The substance which has a fixed chemical composition throughout is called a/an (a) Colloid (b) Inert substance (c) Pure substance (d) Ideal substance.
-
On January 1, 2013, Parflex Corporation exchanged $344,000 cash for 90 percent of Eagle Corporations outstanding voting stock. Eagles acquisition date balance sheet follows: On January 1, 2013,...
-
As a long-term investment, Painters' Equipment Company purchased 20% of AMC Supplies Inc's 470,000 shares for $550,000 at the beginning of the fiscal year of both companies. On the purchase date, the...
-
Using values of f H and S, calculate r G for each of the following reactions at 25C. (a) 2 Pb(s) + O 2 (g) 2 PbO(s) (b) NH 3 (g) + HNO 3 (aq) NH 4 NO 3 (aq) Which of these reactions is (are)...
-
Calculate S(universe) for the formation of 1 mol HCl(g) from gaseous hydrogen and chlorine. Is this reaction spontaneous under standard conditions at 25C? Explain your answer briefly.
-
Consider a French company that pays out 70 percent of its earnings. Its next annual earnings are expected to be 4 per share. The required return for the company is 12 percent. In the past, the...
-
Berset Inc. (BI) has just issued semi-annual coupon bonds with 12 years to maturity and a face value of $1,000 per bond. The bonds make semi-annual coupon payments of $60. What is the price of one...
-
For shooting practice a person uses a pellet gun and an empty pop can. The pop can rests on a flat surface that has a coefficient of kinetic friction 0.500. The pellet has a mass of 0.120 kg and the...
-
Given that 57.7 fex is equal to 1 gal and that 14.3 glo is equal to 1 day, how many cL/hr are there in 0.568 fex/glo? ( NOTE: A fex and glo are are both fictitious units of volume and time,...
-
Category Land Land improvements Buildings Plant Asset $ 165,000 and Amortization $ 1,000,000 219,251 Equipment 625,000 307,500 Automobiles and trucks 162,000 90,325 Leasehold improvements 196,000...
-
137- We want to use K-Means with K=2 for the following data. Where should we initially place the cluster centers? Subject A B 1 1.0 1.0 2 1.5 2.0 34 3 3.0 4.0 4 5.0 7.0 5 3.5 5.0 19 6 4.5 5.0 7 3.5...
-
What is meant by credit risk? To what extent are governments required to disclose the credit risk they have assumed?
-
The time to assemble the first unit on a production line is 10 hours. The learning rate is 0.94. Approximately how long will it take for the seventh unit to be assembled? The number of hours needed...
-
How is it possible to determine the L and S values of a term knowing only the M L and M S values of the states?
-
Can the individual states in Table 22.1 be distinguished experimentally? States and Terms for the np? Configuration TABLE 22.1 M, =m,1 + m,2 Term ML=mn + M2 m;1 m,2 'D -2 1/2 -1/2 -1 3p -1/2 -1/2 1/2...
-
Write an equation giving the relationship between the Rydberg constant for H and for Li 2+ .
-
The three most important questions when conducting an industry analysis. How fast is the industry growing. This is important because is the growth constant or is it something that grew at a fast rate...
-
Petroni Company reports the following selected results for its calendar year 20Y8: Net income $135,000 Depreciation expense 25,000 Loss on sale of land 5,000 Accounts receivable increase 10,000...
-
A9 10-6 C charge is at the origin. At a point (x, y) = (1.8,0) m, what is the magnitude of the electric field due to this charge? N/C At what location x along the x-axis will the electric field equal...

Study smarter with the SolutionInn App


