Cuprite is a semiconductor. Oxide ions are at the cube corners and in the cube center. Copper
Question:
Cuprite is a semiconductor. Oxide ions are at the cube corners and in the cube center. Copper ions are wholly within the unit cell.
(a) What is the formula of cuprite?
(b) What is the oxidation number of copper?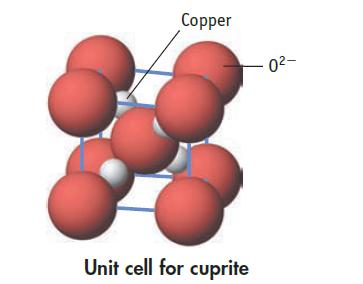
Transcribed Image Text:
Copper Unit cell for cuprite 0²-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
a The formula of cuprite is Cu2O This is because there are two copp...View the full answer

Answered By

Usman Nasir
I did Master of Commerce in year 2009 and completed ACCA (Association of Chartered Certified Accountants) in year 2013. I have 10 years of practical experience inclusive of teaching and industry. Currently i am working in a multinational company as finance manager and serving as part time teacher in a university. I have been doing tutoring via many sites. I am very strong at solving numerical / theoretical scenario-based questions.
4.60+
16+ Reviews
28+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
A particular unit cell is cubic with ions of type A located at the corners and facecenters of the unit cell and ions of type B located at the midpoint of each edge of the cube and at the...
-
Classify each of the following solids as an metallic, molecular, network or ionic solids : Silver (Ag), Silicon Carbide (SiC), Silicon (Si), white phosphorous (P 4 ) Glucose (C 6 H 12 O 6 ),...
-
The structures of another class of high-temperature ceramic superconductors are shown below. a. Determine the formula of each of these four superconductors. b. One of the structural features that...
-
8) Implement a class UserNames which requires an ArrayList of names (user names), consider the following static methods methods: initilizeUserName(), deleteUserName(), addUserName() and display...
-
Evaluate this statement: Good customer service doesnt do you much good, but poor customer service can kill you. Missionary or supporting salespeople do not normally try to secure orders. They are...
-
An investor is holding 1,000 shares of Limited Company. Presently the rate of dividend being paid by company is 2 per share and the share is being sold at 25 per share in the market. However, several...
-
Tests are made on the proportion of defective castings produced by 5 different molds. If there were 14 defectives among 100 castings made with Mold I, 33 defectives among 200 castings made with Mold...
-
Pedro Martinez, who retired from Major League Baseball in 2012, had a stellar career, helping the Boston Red Sox to their first World Series title in 86 years in 2004. The next year he became a free...
-
Use the Chain Rule to find the indicated partial derivatives. z=3x-5y; x = u46v, y = (2u - v); dz dz au av 20 220 = = 12u3-56 (2u-v)3 -24(v-2u)3-48v2 eBook Submit Answer x
-
The mineral fluorite, which is composed of calcium ions and fluoride ions, has the unit cell shown here. (a) What type of unit cell is described by the Ca 2+ ions? (b) Where are the F ions located,...
-
Rutile, TiO 2 , crystallizes in a structure characteristic of many other ionic compounds. How many formula units of TiO 2 are in the unit cell illustrated here? (The oxide ions marked by an x are...
-
In Exercises use the formula for the nth partial sum of a geometric series You go to work at a company that pays $0.01 for the first day, $0.02 for the second day, $0.04 for the third day, and so on....
-
Interview an insurance agent about the differences in cost on a homeowner's or auto insurance policy based on changes in the amount of the deductible that is selected. Write a one-pa.ge report of...
-
For a sample of data where n = 8 given below: a. Calculate the mean, median, and mode. b. Calculate the range, variance, standard deviation, and coefficient of variation. c. Calculate the Z score....
-
Tania Angus established Anguss Phone App Productions in 2018 and kept her accounting records on a cash basis. During 2018, Tania decided to switch her accounting to the accrual basis and has asked...
-
Most mutual fund and brokerage house Internet sites offer free asset allocation and risk-tolerance assessment services. Visit three or four Internet sites and respond to the questions for the asset...
-
Write a list of situations that you can remember as being risky and think about your reaction to each. Do you consider yourself to be a risk-taker or a risk avoider? Now take one of the online risk...
-
Basanti Retirement Home Inc. (Basanti) is a privately owned home for seniors who can live independently but prefer the convenience of having meals, cleaning, and nursing assistance available to them....
-
A manufacturer can sell product 1 at a profit of $20 per unit and product 2 at a profit of $40 per unit. Three units of raw material are needed to manufacture one unit of product 1, and six units of...
-
Draw the alkyl halide that would be necessary to make the amino acid tyrosine using an amidomalonate synthesis. This alkyl halide is highly susceptible to polymerization. Draw the structure of the...
-
When leucine is prepared with an amidomalonate synthesis, isobutylene (also called 2methylpropene) is a gaseous byproduct. Draw a mechanism for the formation of this byproduct.
-
The side chain of tryptophan is not considered to be basic, despite the fact that it possesses a nitrogen atom with a lone pair. Explain.
-
Note: Please make sure to properly format your answers. All dollar figures in the answers need to include the dollar sign and any amount over 1,000 should include the comma ($2,354.67). All...
-
3. Assets A and B are perfectly negatively correlated and have expected returns and standard deviations as follows: Asset A B Expected Return 10 Standard Deviation 8 16 11 What would be the min. you...
-
Mr. Miles obtained a fully amortizing loan of $800,000 at 4% p.a. for 25 years. After 5 years, interest rates have dropped, so that a fully amortizing 20-year loan can be obtained at 3.5% p.a. If the...

Study smarter with the SolutionInn App


