Draw structures for the five possible geometric isomers of Cu(H 2 NCH 2 CO 2 ) 2
Question:
Draw structures for the five possible geometric isomers of Cu(H2NCH2CO2)2(H2O)2. Are any of these species chiral? (See the structure of the ligand in Study Question 78.)
Data given in Question 78
The glycinate ion, H2NCH2CO2−, formed by deprotonation of the amino acid glycine, can function as a bidentate ligand, coordinating to a metal through the nitrogen of the amino group and one of the oxygen atoms.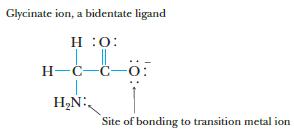
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:





