Review the structure of phosphorous acid in Table 21.6. Data given in Table 21.6 (a) What is
Question:
Review the structure of phosphorous acid in Table 21.6.
Data given in Table 21.6
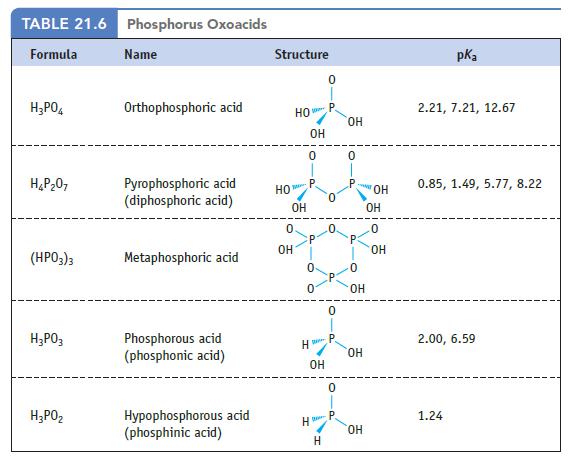
(a) What is the oxidation number of the phosphorus atom in this acid?
(b) Draw the structure of diphosphorous acid, H4P2O5. What is the maximum number of ionizable protons in a molecule of this acid?
Transcribed Image Text:
TABLE 21.6 Phosphorus Oxoacids Formula H3P04 H 207 (HP03)3 H3P03 нарог Name Orthophosphoric acid Pyrophosphoric acid (diphosphoric acid) Metaphosphoric acid Phosphorous acid (phosphonic acid) Hypophosphorous acid (phosphinic acid) Structure НО НО по OH 0 ОН H Mace 0 H ОН OH H 0 0 0 P OH 0 P: ОН OH ОН ОН ОН .0 ОН pka 2.21, 7.21, 12.67 0.85, 1.49, 5.77, 8.22 2.00, 6.59 1.24
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (5 reviews)
a Oxidation number 3 b Dip...View the full answer

Answered By

Rishi Rathore
I was always inclined towards tutoring since my freshman year in college. I started with guiding my younger cousin in pre-calculus and it boosted my self-confidence when he scored a perfect 10 GPA in his school. Later, I went on to join and dedicate two years of time to an NGO named "SAHYOG", which aims at teaching children who come from financially challenged families and cannot afford basic education.
I, along with a team of 3 others, conducted classes of C-Language for juniors in my 4th semester of graduation because of my clarity in C-Programming and ease to interact with students due to my past experiences. I also volunteered for a short period of time for the National Service Scheme (NSS) for underprivileged students and received amazing feedback.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
The single most important concept in the Creative Commons approach is the idea of a spectrum of possibilities between all rights reserved (a conventional copy-right) and no rights reserved (being in...
-
This case study on project evaluation is applicable for beginning courses in corporate finance or finance strategy. Two alternative investment options are available to evaluate. Challenges are...
-
Phosphorous acid, H3PO3, and phosphoric acid, H3PO4, have approximately the same acid strengths. From this information, and noting the possibility that one or more hydrogen atoms may be directly...
-
The following accounts are used by Mouse Potato, Inc., a computer game maker. Required: For each of the following independent situations, give the journal entry by entering the appropriate code(s)...
-
From an economic viewpoint, restrictions on advertising by lawyers look like a device used by the bar to limit competition. (Such advertising restrictions broke down in America, not because they...
-
How do (digital) viruses work?
-
In a dilute solution, the solute obeys (a) Henry's law (b) Raoult's law (c) Dalton's law (d) None of these.
-
Gross Profit Method Sliver Lumber Company handles three principal lines of merchandise with these varying rates of gross profit on cost. Lumber.......................25%...
-
5. Sand is being poured at a rate of 15 in/sec forming an expanding pile. At every instant, the pile of sand is in the shape a cone with equal diameter and height. How fast is the height of the pile...
-
Unlike carbon, which can form extended chains of atoms, nitrogen can form chains of very limited length. Draw the Lewis electron dot structure of the azide ion, N 3 . Is the ion linear or bent?
-
Before hydrazine came into use to remove dissolved oxygen in the water in steam boilers, Na 2 SO 3 was commonly used for this purpose: 2 Na 2 SO 3 (aq) + O 2 (aq) 2 Na 2 SO 4 (aq) What mass of Na 2...
-
For an organization going through an acquisition, would involvement by the internal audit function be considered an assurance or consulting activity? Explain your answer.
-
What is multicollinearity in a regression model?
-
List ten questions you would ask a group of passengers in order to estimate their demand function for a specific airline and a specific route.
-
In a simple regression model, is it possible that all the actual Y values would lie above or below the true regression line? Explain.
-
Write the demand equation, in general form, for an airline and identify the following terms: a. Dependent variable b. Independent variables c. Y-intercept d. Slope e. Logically, one can often expect...
-
Consider the market for jet fuel in a remote regional airport. The domestic demand and supply curves are given as (QS are gallons in thousands): P = 55 3 QD P = 5 + 7 QS. a. What is the market...
-
Samsung Electronics reports the following regarding its accounting for inventories. Inventories are stated at the lower of cost or net realizable value. Cost is determined using the average cost...
-
Tiger, Inc. signed a lease for equipment on July 1, 2007.The lease is for 10 years (the useful life of the asset).The first of 10 equal annual payments of $500,000 was made on July 1, 2007.The...
-
Write an expression for the wave shown in Fig. P.2.43. Find its wavelength, velocity, frequency, and period. Fig. P.2.43 t = 0 60 40 20 z (nm) 100 500 300 -20 -40 -60 t = 0.66 x 10-15 s z (nm) 100...
-
Consider a lightwave having a phase velocity of 3 10 8 m/s and a frequency of 6 10 14 Hz. What is the shortest distance along the wave between any two points that have a phase difference of 30?...
-
Imagine a sound wave with a frequency of 1.10 kHz propagating with a speed of 330 m/s. Determine the phase difference in radians between any two points on the wave separated by 10.0 cm.
-
The matching principle in accounting is a principle that states that companies report expenses the same time as the revenues they are related to. The variable costing differs from this approach in...
-
Using the income statement and the management goals given below, generate a first-draft budget for the upcoming year. Management Goals: Total sales growth of 1.7% Sales mix of 71% food and 29%...
-
Assume Kirtch Company drops Product C. The company then doubles the production and sales of Product B without increasing fixed costs. a. Fill in the chart below to use the total cost approach. After...

Study smarter with the SolutionInn App


