The following photo shows copper balls, immersed in water, floating on top of mercury. What are the
Question:
The following photo shows copper balls, immersed in water, floating on top of mercury. What are the liquids and solids in this photo? Which substance is most dense? Which is least dense? 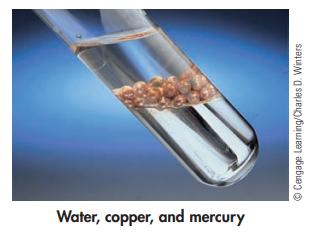
Transcribed Image Text:
Water, copper, and mercury ⒸCengage Learning/Charles D. Winters
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
The liquids in the photo are water and mercury The solids are the co...View the full answer

Answered By

FREDRICK MUSYOKI
Professional Qualities:
Solution-oriented.
Self-motivated.
Excellent problem-solving and critical thinking skills.
Good organization, time management and prioritization.
Efficient troubleshooting abilities.
Tutoring Qualities:
I appreciate students as individuals.
I am used to tailoring resources for individual needs.
I can integrate IT into student's lessons.
I am good at explaining concepts.
I am able to help students progress.
I have a wide curriculum knowledge.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
The table below shows the melting points, boiling points and densities of substances A to D. a. Which substance is a gas at room temperature? b. Which substance is a liquid at room temperature? c....
-
Of all the substances (drugs and alcohol) discussed in the chapter, which substance is most closely connected to crime? Why do you think this is?
-
A solar pond consists of a thin layer of fresh water floating on top of a denser layer of salt water. When the salty layer absorbs sunlight it warms up and much of that heat is held there by the...
-
A 0.831-g sample of SO3 is placed in a 1.00-L container and heated to 1100 K. The SO3 decomposes to SO2 and O2: At equilibrium the total pressure in the container is 1.300 atm. Find the values of Kp...
-
A printing press priced at $315,000 is acquired by trading in a similar press and paying cash for the difference between the trade-in allowance and the price of the new press. a. Assuming that the...
-
In Hollywood movies, it is common to have product placement (having products from sponsored companies, such as cars, appear in movies without telling viewers that these are commercials). As a...
-
Fifty-two percent of the women in the UK play video games regularly. You randomly select seven women in the UK. Find the probability that the number of women in the UK who are gamers is (a) exactly...
-
Clyde had worked for many years as a chef executive of Red Industries INC and had also been a major shareholder. Clyde and the company had a falling out, and Clyde was terminated. Clyde and Red...
-
The cash account for the Justice Company at June 30, 20X8 indicated a balance of $5900. The bank statement indicated a balance of $5800 on June 30, 20X8. Comparing the bank statement and the...
-
Categorize each of the following as an element, a compound, or a mixture. (a). Air (b). Fluorite (c). Brass (d). 18-carat gold
-
Identify the following as either physical changes or chemical changes. (a). The desalination of sea water (separation of pure water from dissolved salts). (b). The formation of SO 2 (an air...
-
Frito-Lay manufactures and markets snack foods. Betsy Gonzalez manages the companys fleet of 200 delivery trucks. Gonzalez has been charged with reengineering the fleet- management function. She has...
-
4) Based on the information provided perform the relevant calculations (EV, PV, AC, CV, SV, CPL, SPI, EAC, ETC) to determine the position of the project. Explain the position of the project. Please...
-
5 slides with the following information Please remember to discuss your learning on each of the following subjects: How to read a credit card statement Calculating credit card interest Credit card...
-
If you win a lottery, you are given two options to collect the prize: taking a $500,000 lump sum today or receiving $100,000 per year for the next ten years. Which would you choose? Please use the...
-
Young Inc. had sales of $545,000 during the year. The company's accounts receivable balance at January 1, was $45,000. Its balance at December 31, the end of the year, is $48,000. What are the...
-
Assume the following data for 136,000 units of a product that Water Source Company expects to produce and sell during the current year: Manufacturing costs: Direct materials $1,471,000 Direct labor...
-
Colorado Mining paid $600,000 to acquire a mine with 40,000 tons of coal reserves. The following statements model reflects Colorado Minings financial condition just prior to purchasing the coal...
-
An interest bearing promissory note for 90 days at 5.6% p.a. has a face value of $120,000. If the note is discounted 20 days after the issue date at a rate of 6.8% p.a., calculate the amount of...
-
Draw the mechanism of the following reaction: ONa + NaBr Br
-
Bromonium ions can be captured by nucleophiles other than water. Predict the products of each of the following reactions: a.
-
For each of the following objects determine whether or not it possesses a plane of symmetry: a. b. c. d. e. f.
-
Compensation is an important element of a high-performance workplace. It is critical to ensure that the compensation philosophy is aligned with the overall organizational strategy. Discuss how a...
-
How can firms employ a portfolio-based approach to strategic resource allocation, optimizing the allocation of capital, talent, and other key resources across diverse business units to maximize...
-
In this section, you will be evaluating various strategies and making recommendations for the Chipotle. Make these recommendations from the perspective of the role of the VP. Write a 1,250- to...

Study smarter with the SolutionInn App


