Use K sp values to decide which compound in each of the following pairs is more soluble.
Question:
Use Ksp values to decide which compound in each of the following pairs is more soluble. (Appendix J.)
(a) AgBr or AgSCN
(b) SrCO3 or SrSO4
(c) AgI or PbI2
(d) MgF2 or CaF2
Data given in Appendix J

Transcribed Image Text:
TABLE 18A Solubility Product Constants at 25 °C Cation Compound Cation Ba²+ *BaCrO Hg₂²+ 2+ BaCO3 BaF₂ *BaSO4 Ca²+ Cut, Cu²+ Au+ Fe²+ Pb²+ Mg²+ Mn²+ CaCO3 (calcite) *CaF2 *Ca(OH)2 CaSO4 CuBr Cul Cu(OH)2 CuSCN AuCl FeCO3 Fe(OH)2 PbBr₂ PbCO3 PbCl₂ PbCrO₁ PbF2 Pbl₂ Pb(OH)2 PbSO4 MgCO3 MgF2 Mg(OH)2 MnCO3 *Mn(OH)2 Ksp 1.2 x 10-10 2.6 × 10-9 1.8 x 10-7 1.1 x 10-10 3.4 x 10-9 5.3 x 10-11 5.5 x 10-5 4.9 x 10-5 6.3 x 10-9 1.3 x 10-12 2.2 x 10-20 1.8 x 10-13 2.0 x 10-13 3.1 x 10-11 4.9 × 10-17 6.6 x 10-6 7.4 x 10-14 1.7 x 10-5 2.8 x 10-13 -13 3.3 x 10-8 9.8 x 10-9 1.4 x 10-15 2.5 x 10-8 6.8 x 10-6 5.2 x 10-11 5.6 x 10-12 2.3 × 10-11 1.9 x 10-13 Ni²+ Ag+ Sr²+ TI+ Zn²+ Compound *Hg₂Br₂ Hg₂Cl₂ *Hg2l2 Hg2SO4 NICO3 Ni(OH)2 *AgBr *AgBrO3 AgCH3CO₂ AgCN Ag₂CO3 *Ag₂C₂04 *AgCl Ag₂ CrO4 *Agl AgSCN *Ag₂SO4 SrCO3 SrF₂ SrSO4 TIBr TICI TII Zn(OH)₂ Zn(CN)₂ Ksp 6.4 x 10-23 1.4 x 10-18 2.9 x 10-29 6.5 x 10-7 1.4 x 10-7 5.5 x 10-16 5.4 x 10-13 5.4 x 10-5 1.9 x 10-3 6.0 x 10-17 8.5 x 10-12 5.4 x 10-12 1.8 x 10-10 1.1 x 10-12 8.5 x 10-17 1.0 x 10-12 1.2 x 10-5 5.6 x 10-10 4.3 × 10-9 3.4 x 10-7 3.7 x 10-6 1.9 x 10-4 5.5 x 10-8 3 x 10-17 8.0 x 10-12
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
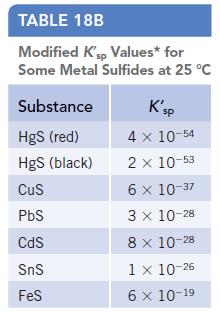
Use K sp values to decide which compound in each of the following pairs is more soluble. (Appendix J.) (a) PbCl 2 or PbBr 2 (b) HgS or FeS (c) Fe(OH) 2 or Zn(OH) 2 Data given in Appendix J TABLE 18A...
-
Which compound in each of the following pairs of compounds is the more soluble one? a. Silver chloride or silver iodide b. Magnesium hydroxide or copper(II) hydroxide
-
Which compound in each of the following pairs would have the higher boiling point? Explain your answers. (a) (b) (c) (d) (e) (f) (g) (h) Hexane, CH3(CH2)4CH3, or nonane, CH3(CH2)7CH3 (i) OH or HO OH...
-
Table P-23 contains Southwest Airlines' quarterly income before extraordinary items ($MM) for the years 1988-1999. a. Plot the income data as a time series and describe any patterns that exist. b. Is...
-
1. Given the facts of this case, should John have been discharged? Explain. 2. Should the sales representatives of AEM be held to a higher standard of personal conduct than sales representatives for...
-
Perceived groups norms, role fulfillment, and status can combine in toxic ways, as a Hydro employee discovered when he was fired in May 2015 for behaviour that occurred off the job at a Toronto...
-
A delicate instrument weighing \(200 \mathrm{~N}\) is suspended by four identical springs, each with stiffness \(50,000 \mathrm{~N} / \mathrm{m}\), in a rigid box as shown in Fig. 9.50. The box is...
-
Vandiver Company had the following select transactions. Apr. 1, 2014 Accepted Goodwin Companys 12-month, 12% note in settlement of a $30,000 account receivable. July 1, 2014 Loaned $25,000 cash to...
-
(14%) Problem 2: A cube is centered on the origin. Its sides are parallel to the ry, the y-z, and the - z planes and have edges of length of L. It is immersed in an electric field given by Refer to...
-
Calculate the molar solubility of silver thiocyanate, AgSCN, in pure water and in water containing 0.010 M NaSCN.
-
If 55 mg of lead(II) sulfate is placed in 250 mL of pure water, does all of it dissolve? If not, how much dissolves?
-
A call option on Illinois stock specifies an exercise price of $38. Today's price of the stock is $40. The premium on the call option is $5. Assume the option will not be exercised until maturity, if...
-
Zack is using the Treynor and Black optimisation method. He has determined that the optimal weighting in the active portfolio is 76.19%. The active portfolio has a residual standard deviation of 5%...
-
What are the concept of learning styles, personalities, and how these concepts are combined with adult learning in organizational training and development programs ?
-
How do the causes of the Great Resignation differ throughout the workforce's various generations?
-
1. What is the work required to raise a 100 kilogram barbell 2 meters upwards? 2. What is the velocity of a 500 kg car that is pulled with 10000 N of force for a total distance of 30 meters? Assume...
-
Alpine Township contracts with Dragoon Environmental Services (DES) to provide solid waste collection to households and businesses. Until recently, DES had an exclusive franchise to provide this...
-
Mary and Nick Stalcheck have an investment portfolio containing 4 investments. It was developed to provide them with a balance between current income and capital appreciation. Rather than acquire...
-
Refrigerant R-12 at 30C, 0.75 MPa enters a steady flow device and exits at 30C, 100 kPa. Assume the process is isothermal and reversible. Find the change in availability of the refrigerant.
-
Explain why two magnetic fields, a static field and a radiofrequency field, are needed to carry out NMR experiments. Why must the two field directions be perpendicular?
-
Explain the difference in the mechanism that gives rise to through-space dipoledipole coupling and through-bond coupling.
-
Predict the number of chemically shifted + 1H peaks and the multiplet splitting of each peak that you would observe for diethyl ether. Justify your answer.
-
What was the government's general approach to budgeting, as outlined in the speech of ministry of finance of Tanzania in 2 0 2 3 / 2 4 ? How did it align with the goals of fiscal responsibility and...
-
3. Critically analyze: a.) The main features and payoff structures of call and put options (3) b.) The determinants of call and put option prices based on Black- Scholes model. (3) c.) How these...
-
2. Explain the techniques of the following two approaches which may be employed by banks to manage interest rate risk. Must provide examples. a.) gap analysis and (5) b.) interest rate swaps (5)

Study smarter with the SolutionInn App


