Use standard enthalpies of formation in Appendix L to calculate enthalpy changes for the following: (a) 1.0
Question:
Use standard enthalpies of formation in Appendix L to calculate enthalpy changes for the following:
(a) 1.0 g of white phosphorus burns, forming P4O10(s)
(b) 0.20 mol of NO(g) decomposes to N2(g) and O2(g)
(c) 2.40 g of NaCl(s) is formed from Na(s) and excess Cl2(g)
(d) 250 g of iron is oxidized with oxygen to Fe2O3(s)
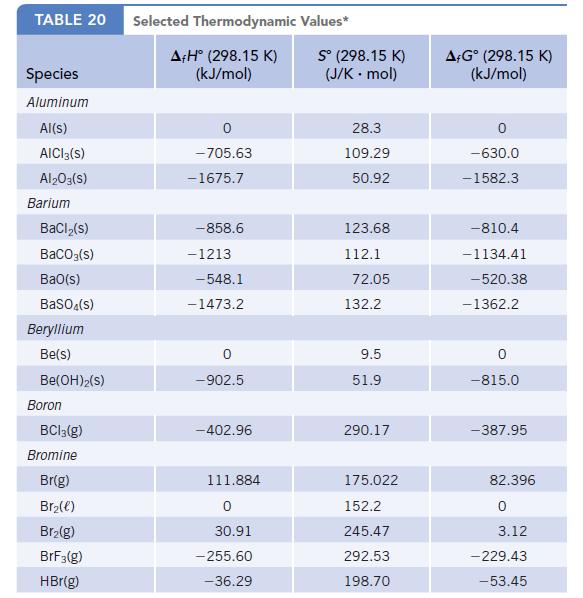
Data given in Appendix L
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:





