What volume of 0.109 M HNO 3 , in milliliters, is required to react completely with 2.50
Question:
What volume of 0.109 M HNO3, in milliliters, is required to react completely with 2.50 g of Ba(OH)2?
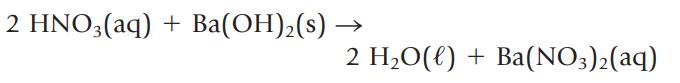
Transcribed Image Text:
2 HNO3(aq) + Ba(OH)₂(s) - 2 H₂O(l) + Ba(NO3)2(aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
To determine the volume of 0109 M HNO3 required to react completely with 250 g of BaOH2 you can use ...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
What volume of 0.750 M Pb(NO 3 ) 2 , in milliliters, is required to react completely with 1.00 L of 2.25 M NaCl solution? The balanced equation is Pb(NO3)2(aq) + 2 NaCl(aq) PbCl(s) + 2 NaNO3(aq)
-
1. What mass of H2 should be produced by the reaction of Al with 75.0 mL of 2.95M HCl? 2Al(s) + 6HCl(aq) 2AlCl3(aq) + 3H2 (g). ln the lab, 0.15g H2 was collected. What is the % yield of the...
-
Marnie wants to save up $250,000 to pay cash for a home purchase 15 years from now. If her investment can carn 6.1% compounded monthly and she intends to grow each payment by 0.25%, what will be her...
-
Suggest two strategies for the company to minimise the impact of the strike on business operation?
-
Describe three recent situations in which you were directly affected by poor product or service quality.
-
If a high-speed spaceship appears shrunken to half its normal length, how does its momentum compare with the classical formula p 5 mv?
-
For each of the following stream functions, with units of \(\mathrm{m}^{2} / \mathrm{s}\), determine the magnitude and the angle the velocity vector makes with the \(x\) axis at \(x=1 \mathrm{~m},...
-
Management Decision Systems (MDS) is a consulting company that specializes in the development of decision support systems. MDS obtained a contract to develop a computer system to assist the...
-
What type of approach is most commonly used for procedures on the digestive organs? Explain why that approach is used.
-
What mass of Na 2 CO 3 , in grams, is required for complete reaction with 50.0 mL of 0.125 M HNO 3 ? NaCO3(aq) + 2 HNO3(aq) 2 NaNO3(aq) + CO(g) + HO(l)
-
What is the hydronium ion concentration of a 1.2 10 4 M solution of HClO 4 ? What is its pH?
-
The Signet Corporation has issued four-month commercial paper with a $6.3 million face value. The firm netted $6,113,898 on the sale. What effective annual rate is Signet paying for these funds?
-
An engineer will deposit 10% of her salary each year into a retirement fund. If her current annual salary is $87,000 and she expects that it will increase by 9% each year, what will be the present...
-
Evans Company reported the following: Manufacturing costs Units manufactured Units sold Beginning inventory $2,385,000 53,000 40,000 units sold for $100 per unit O units What is the amount of gross...
-
Consider a landowner that uses water as a primary input into the production of food. Similar to the problem above, suppose the land has a general production function of f(w) - aw - w, where w is the...
-
If the capital stock is 2 times annual GDP, depreciation is 8% of GDP, and capital income is 20% of GDP, then What is the net marginal product of capital?
-
What audit procedures might you perform to detect fraudulent returns?
-
Davidson Hardware used the FIFO inventory costing method in 2015. Davidson plans to continue using the FIFO method in future years. Which accounting principle is most relevant to Davidsons decision?
-
A bubble-point liquid feed is to be distilled as shown in Figure. Use the Edmister group method to estimate the mole-fraction compositions of the distillate and bottoms. Assume initial overhead and...
-
When benzene is treated with I 2 in the presence of C u Cl 2 , iodination of the ring is achieved with modest yields. It is believed that C u Cl 2 interacts with I 2 to generate I + , which is an...
-
The interior of a refrigerator is typically held at 36F and the interior of a freezer is typically held at 0.00F. If the room temperature is 65F, by what factor is it more expensive to extract the...
-
Using your results from Problem P5.7, calculate q, ÎU, and ÎH for each step in the cycle and for the total cycle described in Figure 5.2. Figure 5.2 Isothermal expansion Pa Adiabatic cold...
-
https://www.youtube.com/watch?v=MJNmbxhKRJQ 1. WAS IT A "PROCESS EVALUATION", OR AN "OUTCOME EVALUATION"? EXPLAIN. 2. DID THE PROGRAM UTILIZE A TRADITIONAL OR PARTICIPATORY APPROACH? EXPLAIN. 3. DID...
-
Solve this case study and provide references and citation as well. case study: Scenario 2: You are a manager at a healthcare organization. Your organization went through changes to leadership...
-
Discuss the various Leadership Styles that are generally implemented by Ranking Officials in the American Law Enforcement Profession. Which Leadership Style(s) do Men generally follow? - Give a...

Study smarter with the SolutionInn App


