Certain elements in the periodic table shown in Figure 7.7 had no electronegativity value defined. Based on
Question:
Certain elements in the periodic table shown in Figure 7.7 had no electronegativity value defined. Based on the definition of electronegativity and the identity of these elements, hypothesize as to why they have no electronegativity value.
Figure 7.7
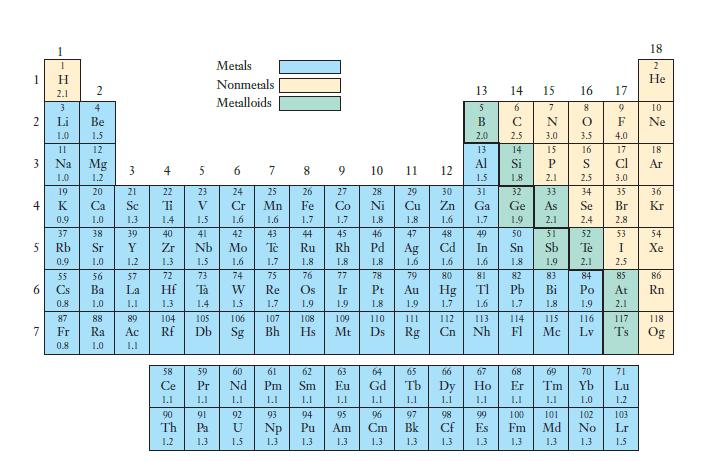
Transcribed Image Text:
1 2 3 4 5 6 7 T H 2.1 3 Li 1.0 11 0.9 55 2 4 Be 1.5 12 Mg 1.2 20 1.0 Ra 1.0 ارا 22 Ti 1.3 1.4 39 Y 1.2 57 La J.L 89 4 23 5 23 V 1.5 40 41 Zr Nh 1.3 1.5 72 73 Ta 1.4 105 Db 1.1 90 91 Th Pa 1.2 1.3 Metals Nonmetals Metalloids. 6 7 24 25 Cr Mn 1.6 1.6 42 43 Mo To 1.6 1.7 74 75 W 15 60 Nd 1.1 92 U 1.3 9 27 Co 542 1.8 76 77 Ir 1.9 10 429 78 Pt 1.8 109 110 Mt Ds 79 1.1 98 CH 1.3 13 5 B 2.0 14 15 6 C 2.5 13 14 Al Si 1.5 1.8 31 32 Ga Ge 1.7 1.9 49 In 1.6 81 F992 67 Ho 1.1 99 Es 13 N 3.0 15 P 2.1 33 1.8 1.9 82 83 Pb Bi 1.7 1.8. 68 Er 1.1 100 Fm 1.3 114 115 Fl Mc 16 8 O 2.4 52 Te 4.0 17 Cl 3.0 35 Br 2.8 53 2.1 2.5 84 85 71 Lu 1.2 103 Lr 1.5 18 118 Og
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Electronegativity is a measure of an atoms ability to attract electrons in a chemical bondIt is a re...View the full answer

Answered By

Aqib Parvej
I am teaching since my graduation time so I have teaching experience of about 5 years and in these years I learn to teach in the best and interesting way .
4.80+
20+ Reviews
41+ Question Solved
Related Book For 

Chemistry For Engineering Students
ISBN: 9780357026991
4th Edition
Authors: Lawrence S. Brown, Tom Holme
Question Posted:
Students also viewed these Sciences questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
The two employees had an altercation on the job site, one used a blunt instrument to inflict a wound on the other employee. the company terminated one employee and suspended the other. The Union...
-
Elements above uranium in the periodic table do not exist in any appreciable amounts in nature because they have short half-lives. Yet there are several elements below uranium in atomic number with...
-
Use the information in Figure to answer the following questions: a. What is the six-month forward rate for the Japanese yen in yen per U.S. dollar? Is the yen selling at a premium or a discount?...
-
What are the six major factors that distinguish multinational financial management from financial management as practiced by a purely domestic firm? Citrus Products Inc. is a medium-sized producer of...
-
What were the differences between German Jewish immigrants and Sephardic Jewish immigrants? Did one group assimilate better than the other? Why?
-
A & R Industrial Supply shows the following financial statement data for 2008, 2009, and 2010. Prior to issuing the 2010 statements, auditors found that the ending inventory for 2008 was understated...
-
You are the chief accountant of Deighton plc, which manufactures a wide range of building and plumbing fittings. It has recently taken over a smaller unquoted competitor, Linton Ltd. Deighton is...
-
(b) Pharoah Company has accounts receivable of $104,500 at March 31, 2022. Credit terms are 2/10, n/30. At March 31, 2022, there is a $1,300 credit balance in Allowance for Doubtful Accounts prior to...
-
How is electronegativity defined?
-
How does the bond energy of a double bond compare to that of two single bonds between the same elements? How does this relationship explain the types of reactions that compounds with double bonds...
-
Wastewater treatment systems are designed to maintain the chemical, physical, and biological integrity of water. These systems, however, tend to generate various greenhouse gases, such as methane...
-
Evaluate the definite integral 5 L (8x+6)dz skmistor
-
Analyze the challenges and techniques involved in process migration across distributed systems. How do operating systems and distributed computing frameworks manage process state, data consistency,...
-
Determine (a) the $ net markdown and (b) the net markdown % from the following figures: Gross Sales $450,000 Customer Returns $10,000 Total Markdowns $124,800 Markdown Cancellation. $32,400
-
Oriole Railroad Co. is about to issue $260,000 of 10-year bonds paying an 10% interest rate, with interest payable annually. The discount rate for such securities is 8%. Click here to view the factor...
-
Explore the security mechanisms in place to isolate processes and prevent unauthorized access to system resources. How do modern operating systems use features like chroot, namespaces, and privilege...
-
You are considering buying a machine that will cost you $12,000. There will be a maintenance cost of $1,000 at the beginning of each year, and the machine will generate cash flows of $5,000 over the...
-
Vectors are drawn from the center of a regular n-sided polygon in the plane to the vertices of the polygon. Show that the sum of the vectors is zero.
-
One of the low-energy geometries of digermane, Ge 2 H 2 , is ethene-like. The Lewis dot structure shown
-
S p hybridization on each Ge atom in planar trans-digermane has been described as sp 1.5 for the GeGe sigma bond and sp 1.8 for the GeH bond. Suppose that the Ge lone electron (in terms of Lewis dot...
-
S p hybridization on each Ge atom in planar trans-digermane has been described as sp 1.5 for the GEGe sigma bond and sp 1.8 for the GeH bond. Calculate the HGeGe bond angle based on this...
-
Need4Speed Bicycle Shop sells specialist racing bicycles. For the purposes of a CVP analysis, the shop owner has divided sales into two categories, as follows: Product type Sales price Cost Sales...
-
1.1 The firm of ICAEW Chartered Accountants where you work is expanding. Partners at the firm are in talks with a number of potential new clients, and the firm has hired several newly qualified ICAEW...
-
Centennial Molding purchased machinery on January 1, 20X1, for $385,000. For 3 years, the company depreciated the machinery using the straight-line method assuming a 12-year useful life and a $24,000...

Study smarter with the SolutionInn App


