Chlorine has only two isotopes, one with mass 35 and the other with mass 37. One is
Question:
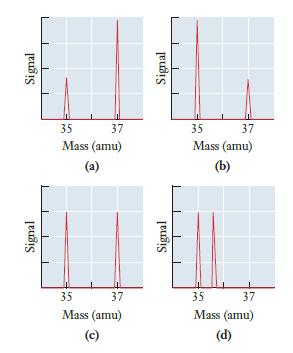
Chlorine has only two isotopes, one with mass 35 and the other with mass 37. One is present at roughly 75% abundance, and the atomic weight of chlorine on a periodic table is 35.45. Which must be the correct mass spectrum for chlorine?
Transcribed Image Text:
Signal Signal 35 37 Mass (amu) (a) 35 37 Mass (amu) (c) Signal Signal T 35 37 Mass (amu) (b) 35 37 Mass (amu) (d)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
b JE...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry For Engineering Students
ISBN: 9780357026991
4th Edition
Authors: Lawrence S. Brown, Tom Holme
Question Posted:
Students also viewed these Sciences questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Mass spectrometry is more often applied to molecules than to atoms. We will see in Chapter 3 that the molecular weight of a molecule is the sum of the atomic weights of the atoms in the molecule. The...
-
A researcher wanted to find out if there was difference between older movie goers and younger movie goers with respect to their estimates of a successful actors income. The researcher first...
-
A student adds NBS to a solution of 1-methylcyclohexene and irradiates the mixture with a sunlamp until all the NBS has reacted. After a careful distillation, the product mixture contains two major...
-
For each case in the accompanying table, answer the questions that follow. a. Calculate the future value of the annuity assuming that it is (1) An ordinary annuity. (2) An annuity due. b. Compare...
-
Micelli Company has an opportunity to invest in one of two projects. Project A requires a $480,000 investment for new machinery with a three-year life and no salvage value . Project B also requires a...
-
Following are observed frequencies. The null hypothesis is H0: p1 = 0.5, p2 = 0.3, p3 = 0.15, p4 = 0.05. a. Compute the expected frequencies. b. Compute the value of X2. c. How many degrees of...
-
Price Company purchased 90% of the outstanding common stock of Score Company on January 1, 2009, for $450,000. At that time, Score Company had stockholders equity consisting of common stock,...
-
A. Compute a 1-day Value at Risk for a bond investment portfolio of K10 million in a long position assuming a 95% confidence interval if the actual daily standard deviation of the portfolio over one...
-
Name each of the following compounds: (a) MgCl 2 , (b) Fe (NO 3 ) 2 , (c) Na 2 SO 4 , (d) Ca (OH) 2 , (e) FeSO 4
-
Based on Figure 3.1, determine (a) The percentage of sunlight that is captured by photosynthesis, (b) The percentage of photosynthesis that occurs in land plants and the percentage that occurs in...
-
United Dominion Manufacturing Co. produces a wood refinishing kit that sells for $17.95. The final processing of the kits occurs in the packaging department. A quilted wrap is applied at the...
-
The equity sections for Atticus Group at the beginning of the year (January Stockholders' Equity (January 1) Common stock-$4 par value, 100,000 shares authorized, 40,000 shares issued and outstanding...
-
Block 1 with mass m is at the origin. Block 2 with mass 1.8 m is a distance d to the right along the x-axis. What is the location on the x-axis of the center of mass of the system? Solve...
-
How can I allow only one word(string) with javax.validation? Which regular expression can I use for @Pattern() I'm not looking for matching the word I want to allow only single word for input with no...
-
Roger files his Federal income tax return 25 days after the due date. Along with the income tax return, he remits a check for $4,000, which is the balance of the income tax owed. The failure to file...
-
You have been analyzing your company's performance over the past few years and decided to conduct a horizontal analysis. 1. Some of the results are causing you concern. What are these and why do they...
-
Adler Industries is a vertically integrated firm with several divisions that operate as decentralized profit centers. Adlers Systems Division manufactures scientific instruments and uses the products...
-
Calculate the Lagrange polynomial P 2 (x) for the values (1.00) = 1.0000, (1.02) = 0.9888, (1.04) = 0.9784 of the gamma function [(24) in App. A3.1] and from it approximations of (1.01) and (1.03).
-
Starting with 1-butanol, show the reagents you would use to prepare each of the following compounds. a. b. c. d. e. H. .
-
Each of the following alcohols can be prepared via reduction of a ketone or aldehyde. In each case, identify the aldehyde or ketone that would be required. a. b. c. d. .
-
Each of the following alcohols can be prepared via reduction of a ketone or aldehyde. In each case, identify the aldehyde or ketone that would be required. a. b. c. d. .
-
Nurse leaders can influence diverse groups of people. This activity is designed to challenge you to think about the opportunities that come with leading and managing a cross-generational workforce...
-
The case study is a Mass Casulty incident fire and the role is Patient Experience Manager at a Hospital Lessons Learned Report Directions. Your Report should address each Aspect requirement as...
-
Ducati made a lot effort (the museum, the "World of Ducati," investment in R&D, etc.) to increase the willingness-to-pay (WTP), and therefore their differentiation advantage, for their products. What...

Study smarter with the SolutionInn App


