Most polymers have densities slightly less than 1 g/cm 3 . How does this compare to the
Question:
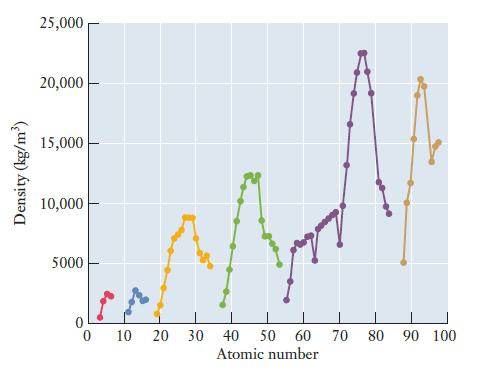
Most polymers have densities slightly less than 1 g/cm3. How does this compare to the densities of metals as shown in Figure 2.11? What does this comparison imply for devices in which conducting polymers might replace metal wires or other components?
Figure 2.11
Transcribed Image Text:
Density (kg/m³) 25,000 20,000 15,000 10,000 5000 0 10 20 30 40 50 60 70 80 90 100 Atomic number
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
To compare the densities we should first convert the given value to the units used in the ...View the full answer

Answered By

Felix Mucee
I am a detailed and thorough professional writer with 5 years of administrative experience- the last 2 years in academic writing and virtual office environment. I specialize in delivering quality services with respect to strict deadlines and high expectations. I am equipped with a dedicated home office complete with a computer, copier/scanner/fax and color printer.
I provide creative and detailed administrative, web search, academic writing, data entry, Personal assistant, Content writing, Translation, Academic writing, editing and proofreading services. I excel at working under tight deadlines with strict expectations. I possess the self-discipline and time management skills necessary to have served as an academic writer for the past five years. I can bring value to your business and help solve your administrative assistant issues.
4.70+
13+ Reviews
33+ Question Solved
Related Book For 

Chemistry For Engineering Students
ISBN: 9780357026991
4th Edition
Authors: Lawrence S. Brown, Tom Holme
Question Posted:
Students also viewed these Sciences questions
-
In our development of consumer theory, we made a big point about the fact that neoclassical economics does not put much stock in the idea of cardinally measuring utility (in terms of units of...
-
The investment banker helping Hologen with the Cybertech acquisition had done some preliminary research and concluded that Hologen could raise $200 million dollars by issuing a 5% coupon bond (paid...
-
The Silicon Valley company Intoot produces checkwriting software. The program itself, Fasten, sells for $50 and includes a package of checks. Check refill packets for Fasten cost $20 to produce and...
-
Consider the following set of data where an experiment was done to determine the molarity of a solute. What mass of sample is needed to reduce the %RSD to 0.10% if the sampling constant is 17.3 g?...
-
Prepare a short report that evaluates each of the following statements separately: 1. Since our plant was shut down for part of the year, we will not depreciate it. Depreciating it for the full year...
-
Hanging from a horizontal beam are nine simple pendulums of the following lengths: (a) 0.10, (b) 0.30, (c) 0.40, (d) 0.80, (e) 1.2, (f) 2.8, (g) 3.5, (h) 5.0, and (i) 6.2 m. Suppose the beam...
-
In September 2013, Ugo Mattera entered into a written construction contract with Baja Properties, LLC. Stephen Chad Golden, the sole owner of Baja Properties, signed the contract and addendums on...
-
Round Tree Manor is a hotel that provides two types of rooms with three rental classes: Super Saver, Deluxe, and Business. The profit per night for each type of room and rental class is as follows:...
-
Using less human effort, less manufacturing space, less investment in tools, and less engineering time is the basic premise of Blank______. Multiple choice question. flexible manufacturing mass...
-
Determine the name of the following ionic compounds: (a) Fe 2 O 3 , (b) Na 2 O, (c) Ca (NO 3 ) 2 Strategy We must determine the names of the constituent ions first. The anions will provide a hint...
-
The fact that a polymers physical properties depend on its atomic composition is very important in making these materials so useful. Why do you think this would be so?
-
John and Martha are planning to be married. Both are professionals each with taxable incomes of $89,700 annually. They are deciding on a wedding date. They have two dates to choose from: December 14,...
-
If a method does not handle a possible checked exception, what must the method have? a . . a throws clause in its header b . . a catch clause in its headerO c . . a finally clause in its header d . ....
-
Which of these can be used to fully abstract a class from its implementation? out of O a . . Objects O b . . None of the Mentioned O c . . Interfaces O d . . Packages
-
k ces CA Qu. 62 What is true about packet sniffers? What is true about packet sniffers? Check All That Apply Legitimate sniffers are used for routine examination and problem detection. Unauthorized...
-
5. Decrypt the ciphertext message "UXDL" that was encrypted with the function f(p) = (5p+ 3) mod 26. ABCDEFGHIJKLM 2 3 4 5 6 7 8 9 10 11 12 0 1 2 NO PQRSTUVWXYZ 13 14 15 16 17 18 19 20 21 22 23 24 25
-
You have been given the following algorithm. What does it return? Input: Array A [ 1 . . . [ 1 . . . n ] ] Output: ? ? ? ? res 1 1 for i 2 2 to n do if A [ [ i ] > ] > A [ [ res ] ] then res i return...
-
Diamonds, Etc manufactures jewelry settings and sells them to retail stores. In the past, most settings were made by hand, and the overhead allocation rate in the prior year was $12 per labor hour...
-
The age-old saying for investing is "buy low and sell high," but this is easier said than done. Investors who panic about falling prices sell their investments, which in turn lowers the price and...
-
Epoxides can be formed by treating α-haloketones with sodium borohydride. Propose a mechanism for formation of the following epoxide. NABH,
-
When methyloxirane is treated with HBr, the bromide ion attacks the less substituted position. However, when phenyloxirane is treated with HBr, the bromide ion attacks the more substituted position....
-
Draw a Lewis structure of a carbon atom that has one extra valence electron (and therefore bears a negative charge). Which second-row element does this carbon atom resemble in terms of the number of...
-
What advanced interventions can organizations implement to address emotional intelligence deficits among employees, such as targeted coaching, training, or immersive experiential learning programs?
-
The nominal interest rate in the U.S. is 5% and the nominal interest rate in Canada is 3%. The spot value of the U.S. dollar is 1 ($/Canadian dollar) and the forward rate is 1.2 ($/Canadian dollar)....
-
2. Two players play the following game for infinite times. Cooperate Betray Cooperate 10, 20 15,-22 Betray -25,30 -12,-18 For the player to continue to cooperate what would be the ranges of their...

Study smarter with the SolutionInn App


