Suppose that speed distributions for each of the following gases were added to Figure 5.7. Which of
Question:
Suppose that speed distributions for each of the following gases were added to Figure 5.7. Which of the four gases in the original figure would each most closely resemble? Explain your answer.
(a) Ar
(b) Ne
(c) CH4
Figure 5.7
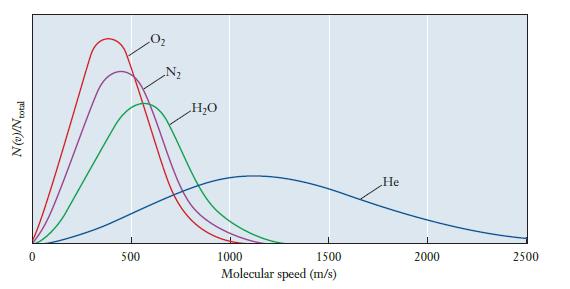
Transcribed Image Text:
N(v)/N total 0 500 N₂ H₂O 1000 1500 Molecular speed (m/s) He 2000 2500
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
Because temperature is fixed the ...View the full answer

Answered By

Pushpinder Singh
Currently, I am PhD scholar with Indian Statistical problem, working in applied statistics and real life data problems. I have done several projects in Statistics especially Time Series data analysis, Regression Techniques.
I am Master in Statistics from Indian Institute of Technology, Kanpur.
I have been teaching students for various University entrance exams and passing grades in Graduation and Post-Graduation.I have expertise in solving problems in Statistics for more than 2 years now.I am a subject expert in Statistics with Assignmentpedia.com.
4.40+
3+ Reviews
10+ Question Solved
Related Book For 

Chemistry For Engineering Students
ISBN: 9780357026991
4th Edition
Authors: Lawrence S. Brown, Tom Holme
Question Posted:
Students also viewed these Sciences questions
-
1. How strong are the competitive forces confronting J. Crew in the market for specialty retail? Do a [Michael Porter] five-forces analysis to support your answer. (see chapter 3 in the textfor...
-
KYC's stock price can go up by 15 percent every year, or down by 10 percent. Both outcomes are equally likely. The risk free rate is 5 percent, and the current stock price of KYC is 100. (a) Price a...
-
Explain the meaning of the terms emoluments, employments and office for the purposes of PAYE as you earn systems. 2. Explain the actual receipts basis of assessing the emoluments from the employment...
-
A 14-foot piece of string is cut into two pieces so that the longer piece is 2 feet longer than twice the shorter piece. Find the lengths of both pieces. What is the lenath of the shorter oiece?1...
-
Derby Company prepares monthly cash budgets. Relevant data from operating budgets for 2013 are: All sales are on account. Collections are expected to be 60% in the month of sale, 30% in the first...
-
The Hydro-Electric Company (HEC) has three sources of power. A small amount of hydroelectric power is generated by damming wild and scenic rivers: a second source of power comes from burning coal,...
-
What is the scope of the discovery?
-
Heavenly Displays Inc. puts together large-scale fireworks displaysprimarily for Canada Day celebrations sponsored by corporations and municipalities. The company assembles and orchestrates complex...
-
7. (1 point) Express the function y = x+6 as a composition y = f(g(x)) of two simpler functions y = f(u) and u = g(x). Generated by WeBWorK, http://webwork.maa.org, Mathematical Association of...
-
Why do heavier gases move more slowly than light gases at the same temperature?
-
Use the kinetic theory to explain what happens to the pressure exerted by a gas as its temperature is increased.
-
Strategic analysis of operating income (continuation of 13-22) Refer to Exercise 13-22. 1. Calculate the operating income of Meredith Corporation in 2008 and 2009. 2. Calculate the growth,...
-
Suppose you walk into a sauna that has an ambient temperature of 55 degrees celsius (a) Calculate the rate of heat transfer to you by radiation in watts, given your skin temperature is 39 degrees...
-
Mary Burden is the CFO of Tidewell Corporation and Tommy Brown is the Treasurer. Mary has been with the company for seven years and Tommy just started earlier this year. They meet to discuss the...
-
Calculate the velocity of sound in nitrogen gas at 25C. For this problem, assume that nitrogen is an ideal gas. b) (2 points) How does your calculated value compare to the often-cited value for the...
-
Department G had 1,920 units 25% completed at the beginning of the period, 12,200 units were completed during the period, 1,600 units were 20% completed at the end of the period, and the following...
-
Find the derivative of y with respect to the given independent variable. y=log 36ex-log6 x dy dx 11
-
Using Figure 9.2 as a guide, name three internal and three external databases that you think should be included in a data warehouse for your university. Support your answer.
-
A copper sphere of 10-mm diameter, initially at a prescribed elevated temperature T;, is quenched in a saturated (1 atm) water bath. Using the lumped capacitance method, estimate the time for the...
-
The amplitude of a pendulum consisting of a mass on a long wire is initially adjusted to have a very small value. The amplitude is found to decrease slowly with time. Is this process reversible?...
-
A process involving an ideal gas is carried out in which the temperature changes at constant volume. For a fixed value of T, the mass of the gas is doubled. The process is repeated with the same...
-
You are told that S = 0 for a process in which the system is coupled to its surroundings. Can you conclude that the process is reversible? Justify your answer.
-
The bars AB and AC are joined by a pin at A and a horizontal cable. The vertical cable carrying the 200 kg mass is attached to the pin at A. Determine the tension in the horizontal cable. Neglect the...
-
1. The 25-kg box is launched from the position shown along the rough horizontal plane with the velocity of 8 m/s. Determine the distance x that the box will travel before the spring stops forward...
-
Calculate the maximum stress in a 5 mm thick steel plate with an 1 8 mm diameter round hole, given h gross = 1 2 cm and the tensile load is 8 . 5 kN . ( Final answer in MPa ) P h gross d

Study smarter with the SolutionInn App


