A compound with the formula C 5 H 8 could have either of the following structures. If
Question:
A compound with the formula C5H8 could have either of the following structures. If the addition of an equimolar amount of molecular hydrogen yields only a single pure compound, which structure is likely to be correct?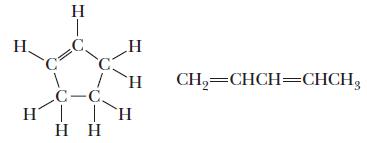
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
The structure on the left is likely to be correctThis is because the structure on th...View the full answer

Answered By

Akshay Singla
as a qualified engineering expert i am able to offer you my extensive knowledge with real solutions in regards to planning and practices in this field. i am able to assist you from the beginning of your projects, quizzes, exams, reports, etc. i provide detailed and accurate solutions.
i have solved many difficult problems and their results are extremely good and satisfactory.
i am an expert who can provide assistance in task of all topics from basic level to advance research level. i am working as a part time lecturer at university level in renowned institute. i usually design the coursework in my specified topics. i have an experience of more than 5 years in research.
i have been awarded with the state awards in doing research in the fields of science and technology.
recently i have built the prototype of a plane which is carefully made after analyzing all the laws and principles involved in flying and its function.
1. bachelor of technology in mechanical engineering from indian institute of technology (iit)
2. award of excellence in completing course in autocad, engineering drawing, report writing, etc
4.70+
48+ Reviews
56+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Create an accompanying report that draws on relevant theories and concepts within Block 1 to explain and justify the decisions made in producing your team's piece of marketing communication. Context...
-
Although the death penalty in India has similarities to the United States, the Indian Supreme Court and the Presidents of India have successfully halted nearly all executions. How have t he Supreme...
-
Perform the same computation as Sec. 24.1, but compute the amount of heat requited to raise the temperature of 1200 g of the material from -150 to 100C. Use Simpsons rule for your computation, with...
-
The inductor of Problem 1.13 is to be operated from a 60-Hz voltage source. (a) Assuming negligible coil resistance, calculate the rms inductor voltage corresponding to a peak core flux density of...
-
If two orders are selected, find the probability that they are both from Taco Bell. a. Assume that the selections are made with replacement. Are the events independent? b. Assume that the selections...
-
If you were trying to examine this issue, how would you proceed? Any suggestions for me to take next steps? same. Is that what happened? You normally wouldnt borrow money from the company, would you?
-
Danish company Bang & Olufsen (B&O) is known globally for its high-end audio and video. CEO Kalle Hvidt Nielsen says, Our mission is to make complex technology very simple to use. B&O uses contract...
-
How much energy does a male Jackson's widowbird of mass 45 g expend when jumping to a height of 1.0 m during its mating ritual? Neglect all losses due to air resistance
-
Suppose the Scope Triangle were modified as follows: Resource Availability occupies the center, and the three sides are Scope, Cost, and Schedule. Interpret this triangle as if it were a system in...
-
Describe the most important intermolecular force between molecules of (a) 2-methyl-2-propanol. (b) 2-butanone.
-
Let W be the set of all vectors of the form shown, where a, b, and c represent arbitrary real numbers. In each case, either find a set S of vectors that spans W or give an example to show that W is...
-
Suppose you buy a May expiration put option on 100 shares with the exercise price of $50. If the stock price at option expiration is $47, will you exercise your put? What is the net profit/loss on...
-
Johnson and Company had a balance in their retained earnings account at the end of 2022 in the amount of 1,500,000. They have forecasted net income in 2023 in the amount of 400,000. They pay an...
-
This semester, we've been talking about three of the four principles of ethics: autonomy, nonmaleficence, and beneficence. In this assignment, we're going to discuss the concept of justice. 1. What...
-
How would you manage termination of employment (aka separation) from this job? There are some general steps that are applicable to separation from any job, but are there any unique aspects of...
-
Raj works at an upscale department store. Because of his religion, Raj regularly wears a headscarf. Several customers and employees complain to management that they feel uncomfortable around Raj...
-
During August 2010, Berman Companys Department Y equivalent unit product costs, computed under the weighted average method, were as follows: Material .........$2 Conversion ....... 6 Transferred in...
-
Inexhaustible collections of ONPOs are not required to be capitalized or depreciated, if certain criteria are met. Why is this so, and what accounting and reporting recognition, if any, is required...
-
When magnesium is placed in an acid solution, hydrogen gas is given off. Is magnesium or hydrogen the better reducing agent?
-
A displacement reaction is an oxidation-reduction reaction in which one element displaces another from solution. In each of the following displacement reactions identify the element that is oxidized...
-
Hydrogen sulfide gas dissolves in water and dissociates very slightly: H 2 S 2H + + S 2 -. How would the acidity of the solution be affected by a. Increasing the pressure of H 2 S? b. Raising the...
-
Current Attempt in Progress Carla Vista's Corner Store is adding gasoline pumps with underground tanks to store the fuel. The tanks have an estimated life of 30 years. Carla Vista's estimates that...
-
What is the eumber of ights needed to purchafe one thinevsshare? Venture Berhad plans to raise RM20 million from a rights issue to fund a new business expansion. The company will issue 8 million new...
-
Required information [The following information applies to the questions displayed below.] Benton Corporation manufactures computer microphones, which come in two models: Standard and Premium. Data...

Study smarter with the SolutionInn App


