A mixture of antimony atoms and Cl 2 in the gas phase reacts as shown in the
Question:
A mixture of antimony atoms and Cl2 in the gas phase reacts as shown in the drawing below.
(a) Write the balanced equation.
(b) Which reactant is the limiting reactant?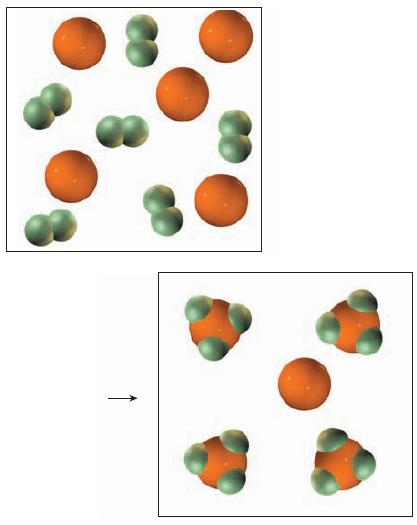
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
a Balanced Equation The reaction between antimony atoms and chlorine gas produces antimony trichlori...View the full answer

Answered By

Sarfraz gull
have strong entrepreneurial and analytical skills which ensure quality tutoring and mentoring in your international business and management disciplines. Over last 3 years, I have expertise in the areas of Financial Planning, Business Management, Accounting, Finance, Corporate Finance, International Business, Human Resource Management, Entrepreneurship, Marketing, E-commerce, Social Media Marketing, and Supply Chain Management.
Over the years, I have been working as a business tutor and mentor for more than 3 years. Apart from tutoring online I have rich experience of working in multinational. I have worked on business management to project management.
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
As society searches for technical solutions to global warming, one approach to sequester carbon-dioxide rich greenhouse gases is to capture the CO 2 within an adsorbent material at high pressure. An...
-
A stirred tank with volume V t? (L) is charged with V 1 (L) of a liquid. B. The space above the liquid (volume V g = V t ? V 1 ) is filled with a pure gas, A, at an initial pressure P 0 (atm). The...
-
You have two properties to refinance. Property A is an office building that generates $325,000 in NOI and is worth $5,000,000. Property B is a warehouse that generates $425,000 in NOI and is worth...
-
1. Matching Match the graph of the rational function f (x) = (ax + b) / (cx + d) with the given conditions. (a) (b) (c) (d) (i) a > 0 (ii) a > 0 (iii) a 0 b 0 b > 0 b c > 0 c 0 c > 0 d 0
-
A pendulum is constructed from two identical uniform thin rods a and b each of length L and mass m, connected at right angles to form a T by joining the centre of rod a to one end of rod b. The T is...
-
Find an appropriate graphing software viewing window for the given function and use it to display its graph. The window should give a picture of the overall behavior of the function. There is more...
-
DJ Slick was a radio show host on WLTO-FM, based in Lexington, Kentucky. As a promotional, he announced that he would reward loyal listeners with a chance to win one hundred grand by calling the...
-
City Place Movie Theaters has four employees and pays them on an hourly basis. During the week beginning June 24 and ending June 30, 2016, these employees worked the hours shown below. Information...
-
1 2 34 What is "SWOT analysis? How do you carry it for a technical educationalinstitute? What is corporate planning? Explain the process of corporate planning? Discuss the process of strategy...
-
Redang Chalet Sdn Bhd (RCSB) rents out chalet to tourists at a popular beach in Redang, Terengganu. RCSB own 10 chalets, in which the construction cost amounted to RM1 million. Each chalet is...
-
Calculate the mass of silver produced if 3.22 g zinc metal and 4.35 g AgNO 3 react according to the following equation. Zn(s) + 2AgNO3(aq) 2Ag(s) + Zn(NO3)2(aq)
-
In a reaction of HCl and NaOH, the theoretical yield of H 2 O is 78.2 g. What is the theoretical yield of NaCl?
-
Which statement below is correct? a. Real property is depreciated using the half-year convention. b. Residential real estate is depreciated over a 39-year life. c. One-half month of depreciation is...
-
How would you set up crosses to determine if a gene is Y-linked versus X-linked?
-
Mexican hairless dogs have little hair and few teeth. When a Mexican hairless is mated to another breed of dog, about half of the puppies are hairless. When two Mexican hairless dogs are mated to...
-
Charlie is hiking along a trail on forested land in the state of Maine near the United States border with Canada. Beside the trail, Charlie finds a backpack that contains $100,000 in cash. He reports...
-
The process of binary fission begins with a single mother cell and ends with two daughter cells. Would you expect the mother and daughter cells to be genetically identical? Explain why or why not.
-
Xeno Corporation purchased supplies from Kimbo Company in 2018. The total invoice was for \(\$ 20,000\), but Xeno claimed that only one-half of the order was received and paid only \(\$ 10,000\) in...
-
A company manufactures a product at two plants, one in Dallas and one in Kansas City. The Dallas plant can produce up to 200 units per day, and the Kansas City plant can produce up to 250 units per...
-
Consider the discrete group G of order 8 that has the following Cayley diagram e If we have the sequence of operations: fcagec, which of the options represents the reduction of the sequence to a...
-
When a nucleophile and electrophile are tethered to each other (that is, both present in the same compound), an intramolecular substitution reaction can occur, as shown. Assume that this reaction...
-
For the substitution reaction shown below, assume a stepwise process is taking place and draw the mechanism. + NaCI NaBr Br CI
-
The reaction below exhibits a second-order rate equation: (a) What happens to the rate if the concentration of 1-iodopropane is tripled and the concentration of sodium hydroxide remains the same? (b)...
-
Froya Fabrikker A/S of Bergen, Norway, manufactures specialty heavy equipment for use in North Sea oil fields. The company uses a job-order costing system that applies manufacturing overhead cost to...
-
O'Riley Company's payroll costs and fringe benefit expenses include the normal CPP and El contributions, retirement fund contributions of 10% of total earnings, and health insurance premiums of $131...
-
1) Construct control charts for ranges and means using the following table. Are any samples beyond the control limits? Size of Sample (n) Factor for UCL and LCL for Factor for LCL for Factor for UCL...

Study smarter with the SolutionInn App


