A molecular model of methyl alcohol is shown below; the OH group is an alcohol functional group,
Question:
A molecular model of methyl alcohol is shown below; the OH group is an alcohol functional group, a group present in many important chemicals. Write the formula of methyl alcohol and use it to calculate the number of moles of hydrogen atoms in 0.33 mol methyl alcohol.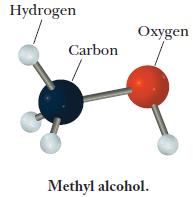
Transcribed Image Text:
Hydrogen Carbon Oxygen Methyl alcohol.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
CH ...View the full answer

Answered By

Muhammad Ahtsham Shabbir
I am a professional freelance writer with more than 7 years’ experience in academic writing. I have a Bachelor`s Degree in Commerce and Master's Degree in Computer Science. I can provide my services in various subjects.
I have professional excellent skills in Microsoft ® Office packages such as Microsoft ® Word, Microsoft ® Excel, and Microsoft ® PowerPoint. Moreover, I have excellent research skills and outstanding analytical and critical thinking skills; a combination that I apply in every paper I handle.
I am conversant with the various citation styles, among them; APA, MLA, Chicago, Havard, and AMA. I also strive to deliver the best to my clients and in a timely manner.My work is always 100% original. I honestly understand the concern of plagiarism and its consequences. As such, I ensure that I check the assignment for any plagiarism before submission.
4.80+
392+ Reviews
587+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Shown in the margin is one molecule of methylamine; the NH 2 group is an example of an amine functional group, a group present in many important chemicals. Write the formula of methylamine and use it...
-
A molecular model of hydrogen peroxide is shown below. Write the formula of hydrogen peroxide and use it to calculate the number of moles of hydrogen atoms in 0.011 mol hydrogen peroxide. Hydrogen...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
In each of the homeowners forms, OA) the property coverage is the same OB) the liability coverage varies OC) both the property and liability coverage are the same OD) the property coverage varies
-
A 4-cm-square waveguide is filled with a dielectric with complex permittivity c = 16 o (l j10 4 ) and is excited with the TM 21 mode. If the waveguide operates at 10% above the cutoff frequency,...
-
A(5, 5), B(3, 7), C(12, 3), and D(3, 6) are four points. The lines AB and CD meet at P. Find the coordinates of P.
-
In 1997, Bernard Bilski and Rand Warsaw filed a patent application for a method of hedging risk in the field of commodities trading. In effect, the asserted invention was a method of protecting...
-
Ramirez Company installs a computerized manufacturing machine in its factory at the beginning of the year at a cost of $ 43,500. The machines useful life is estimated at 10 years, or 385,000 units of...
-
6. An igloo, a hemispherical enclosure built of ice (1.67 J/m-sC), has as inner radius of 2.50m. The thickness of the ice is 0.5m. At what rate must thermal energy be generated to maintain the air...
-
The following is Edge Sports Repair Shop's trial balance at September 30, 2014, the company's fiscal year end: Additional information: 1. Service revenue earned but not recorded at September 30,...
-
What is the percentage, by mass, of each element in the following substances? (a) C 4 H 8 (b) C 3 H 4 N 2 (c) Fe 2 O 3
-
Nickel tetracarbonyl, Ni(CO) 4 , is a volatile (easily converted to the gas phase), extremely toxic compound that forms when carbon monoxide gas is passed over finely divided nickel. Despite this...
-
Julie Ellis asks your help concerning an NSF check. Explain to Julie (a) What an NSF check is, (b) How it is treated in a bank reconciliation, and (c) Whether it will require an adjusting entry on...
-
Let \(y\) follow a mixture of structural zeros of probability \(p\) and a Poisson distribution with mean \(\mu\) of probability \(q=1-p\). Show that \(E(y)=q \mu\), and \(\operatorname{Var}(y)=q...
-
Suppose you test ten hypotheses and under the null hypothesis each hypothesis is to be rejected with type I error rate 0.05. Assume that the hypotheses (test statistics) are independent. Compute the...
-
Show that the odds ratio is a monotone function of \(p_{11}\) if marginal distributions are fixed.
-
Let \(\mathbf{x}\) be a random vector and \(\boldsymbol{V}\) its variance matrix. Show that \(\mathbf{x}^{\top} \boldsymbol{V}^{-1} \mathbf{x}\) is invariant under linear transformation. More...
-
Use the DOS data to test whether there is gender and depression (dichotomized according to no and minor/major depression) association by stratifying medical burden and education levels, where medical...
-
Jerilu Markets has a beta of 1.09. The risk-free rate of return is 2.75 percent and the market rate of return is 9.80 percent. What is the risk premium on this stock? A. 6.47 percent B. 7.03 percent...
-
What are the risks and liability factors in an audit? What are the implications to the auditor? What are the implications to the organization? How can the auditor mitigate these risks and liability...
-
An ideal gas sample containing 1.75 moles for which C V ,m = 5/2R undergoes the following reversible cyclical process from an initial state characterized by T = 275 K and P = 1.00 bar: a. It is...
-
For protein denaturation, the excess entropy of denaturation is defined as is the transition excess heat capacity. The way in which δC trs P can be extracted from differential scanning...
-
The standard entropy of Pb(s) at 298.15 K is 64.80 J K -1 mol - 1 . Assume that the heat capacity of Pb(s) is given by The melting point is 327.4C and the heat of fusion under these conditions is...
-
The partnership agreement of Jones, King, and Lane provides for the annual allocation of the business's profit or loss in the following sequence: Jones, the managing partner, receives a bonus equal...
-
How many pairs of positive integers x and y satisfy the equation x + 2y = 210?
-
Let be the arbitrage-free forward exchange rate on a currency forward contract with time to expiration (T t) and be the value of a forward exchange contract with a strike of (i.e. a contract to buy...

Study smarter with the SolutionInn App


