A molecular model of hydrogen peroxide is shown below. Write the formula of hydrogen peroxide and use
Question:
A molecular model of hydrogen peroxide is shown below. Write the formula of hydrogen peroxide and use it to calculate the number of moles of hydrogen atoms in 0.011 mol hydrogen peroxide.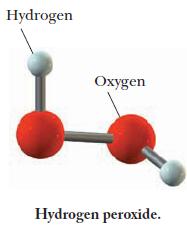
Transcribed Image Text:
Hydrogen Oxygen Hydrogen peroxide.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
The formula of hydrogen peroxide is HO As you can see from the structural formula there are tw...View the full answer

Answered By

Amar Kumar Behera
I am an expert in science and technology. I provide dedicated guidance and help in understanding key concepts in various fields such as mechanical engineering, industrial engineering, electronics, computer science, physics and maths. I will help you clarify your doubts and explain ideas and concepts that are otherwise difficult to follow. I also provide proof reading services. I hold a number of degrees in engineering from top 10 universities of the US and Europe.
My experience spans 20 years in academia and industry. I have worked for top blue chip companies.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
A molecular model of methyl alcohol is shown below; the OH group is an alcohol functional group, a group present in many important chemicals. Write the formula of methyl alcohol and use it to...
-
The decomposition of hydrogen peroxide is catalyzed by iodide ion. The catalyzed reaction is thought to proceed by a two-step mechanism: H2O2s(aq) + I (aq) H2O(I) + IO (aq) (slow) IO (aq) + H2O2(aq)...
-
Write a structural formula or build a molecular model of each of the following: (a) 1-Octyne (b) 2-Octyne (c) 3-Octyne (d) 4-Octyne (e) 2,5-Dimethyl-3-hexyne (f) 4-Ethyl-1-hexyne (g)...
-
According to the Statute of Frauds, in order to be legally enforceable, a contract must be in writing, name the contracting parties, identify the subject matter of the contract, and Be for a legal...
-
If the walls of the square waveguide in the previous problem are made of brass (ac = 1.5 107 S/m), find ac and the distance over which the wave is attenuated by 30%.
-
The diagram shows triangle ABC with AB perpendicular to BC. A is the point (6, 6) and B is the point (5, 2). The gradient of AC is 2/9. Find the coordinates of C. A(-6, 6) B(-5,2) YA X
-
Trade secret theft can cause significant harm to a business. At the same time, businesses sometimes intentionally suppress information that might harm sales if publicly known. Suppose an employee of...
-
The records of Romano Inc. reflect the following data: Work in process, beginning of month 2,000 units one-half completed at a cost of $1,250 for materials, $675 for labor, and $950 for overhead....
-
7. A vertical spring is fixed to one of its end and a massless plank fitted to the other end. A block is released from a height has shown. Spring is in relaxed position. Then choose the correct...
-
Hotel DelRay is located at the heart of the city of Brussels, in Belgium. Brussels is a major hub for international politics, a home for several international organizations and diplomats, and a...
-
What is the percentage, by mass, of each element in the following substances? (a) C 4 H 8 (b) C 3 H 4 N 2 (c) Fe 2 O 3
-
Nickel tetracarbonyl, Ni(CO) 4 , is a volatile (easily converted to the gas phase), extremely toxic compound that forms when carbon monoxide gas is passed over finely divided nickel. Despite this...
-
Testing to see if there is evidence that the proportion of US citizens who can name the capital city of Canada is greater than 0.75. Use the following possible sample results: Sample A: 31 successes...
-
Prove that the CMP distribution \(\operatorname{CMP}(\lambda, v)\) converges to (a) a Bernoulli distribution as \(v\) goes to infinite. Find the parameter for the limiting Bernoulli distribution; (b)...
-
Each of the three random variables \(x, y\), and \(z\) has two levels: 0 and 1 . The joint distribution of these three variables can be determined from the facts \(\operatorname{Pr}(x=0, y=\) \(0,...
-
Have you experienced Simpson's paradox in your professional and/or personal life? If so, please describe the context in which it occurred.
-
Use the log-linear model to test whether SCID (two levels: no depression and depressed including major and minor depression) and dichotomized EPDS (EPDS \(\leq 9\) and EPDS \(>9)\) are homogeneously...
-
Assess the models in Problems 9.12 and 9.14 Problem 9.12 is: and Problem 9.14 is: . 9.12 Generalize the model considered in Example 4.11 to a marginal model for the longi- tudinal DOS data and...
-
What is the standard deviation of the returns on a stock given the following information? A. 1.57 percent B. 2.03 percent C. 2.89 percent D. 3.42 percent E. 4.01percent Probablity of at f Return...
-
In your readings, there were many examples given for nurturing close family relationships in this ever-evolving technological society we live in Based upon your readings and research describe three...
-
Propose an efficient synthesis for the following transformation. Br
-
The mean solar flux at the Earths surface is ~2.00 J cm 2 min 1 . In a non-focusing solar collector, the temperature reaches a value of 79.5C. A heat engine is operated using the collector as the hot...
-
Propose a plausible mechanism for the following transformation. C CI
-
The Distance Plus partnership has the following capital balances at the beginning of the current year along with respective profit and loss percentages: Tiger (50%) Phil (20%) Ernie (30%) $ 135,000...
-
my work Lamothe Kitchen and Bath makes products for the home, which it sells through major retailers and remodeling (do-it-yourself, or DIY) outlets. One product that has had varying success is a...
-
The Prince-Robbins partnership has the following capital account balances on January 1, 2024: Prince, Capital Robbins, Capital $ 165,000 155,000 Prince is allocated 60 percent of all profits and...

Study smarter with the SolutionInn App


